PSA provocation by bipolar androgen therapy may predict duration of response to first-line androgen deprivation: Updated results from the BATMAN study
- PMID: 35938545
- PMCID: PMC9633380
- DOI: 10.1002/pros.24426
PSA provocation by bipolar androgen therapy may predict duration of response to first-line androgen deprivation: Updated results from the BATMAN study
Abstract
Background: Previously, we reported results from the Phase II BATMAN study (Bipolar Androgen Therapy for Men with Androgen-ablation Naïve prostate cancer). This study (NCT01750398) was designed to evaluate the safety and efficacy of a treatment regimen consisting of a 6-month lead in-phase of androgen deprivation therapy (ADT) followed by alternating 3-month intervals of bipolar androgen therapy (BAT) and ADT alone. Here we report > 5-year follow-up related to the duration of subsequent ADT, response to first-line androgen receptor inhibitors, safety, and survival in men with castration-sensitive prostate cancer treated on the BATMAN study.
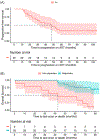
Methods: Univariate Cox regression was utilized to compare overall survival between Responders who achieved a prostate-specific antigen (PSA) level of <4 ng/ml and Non-Responders who achieved a PSA level of ≥4 ng/ml after BAT/ADT. Kaplan-Meier method and Cox regression were used to assess progression-free (PFS) and overall survival (OS) on BAT and on subsequent abiraterone or enzalutamide and on the association between PSA peak during BAT and each time to event outcome.
Results: Over median follow-up of 95 months, the median PFS on ADT for the entire cohort was 47.8. Median OS has not been reached (NR). Median OS for Non-Responders is 43 months versus NR (not reached) for responders (hazard ratio [HR]: 0.176, p = 0.002). Post-BAT, the PSA50 and PSA90 responses to abiraterone or enzalutamide were 94.4% and 66.7%, respectively and median PFS was 20.6 months. Patients with peak PSA level of ≥9 ng/ml after BAT had median PFS of 20.6 months versus NR for those having PSA < 9 ng/ml (HR: 0.122, p < 0.001). Median OS was 79.6 months for patients with PSA peak of ≥9 ng/ml versus NR for those having PSA peak of <9 ng/ml (HR: 0.409, p = 0.131).
Conclusion: The use of BAT as part of first-line hormonal therapy strategy does not induce adversely affect long-term survival or induce any significant long-term adverse sequelae in patients with prostate cancer. Cycling BAT may extend the duration of ADT response and enhance response to subsequent androgen ablative therapies. The magnitude of BAT-provoked increase in PSA may predict duration of ADT response and warrants further study.
Keywords: PSA; androgen deprivation; bipolar androgen therapy; castration-sensitive.
© 2022 Wiley Periodicals LLC.
Conflict of interest statement
CONFLICT OF INTEREST
The authors declare no conflict of interest.
Figures




References
-
- Huggins C, Hodges CV. Studies on prostatic cancer. I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. J Urol. 2002;167:948–951. - PubMed
-
- Schweizer M, Wang H, Luber B, et al. Bipolar androgen therapy for men with androgen ablation naïve prostate cancer: results from the phase II BATMAN study. Prostate. 2016;76:1218–1226. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

