Succinate dehydrogenase and MYC-associated factor X mutations in pituitary neuroendocrine tumours
- PMID: 35938916
- PMCID: PMC9513646
- DOI: 10.1530/ERC-22-0157
Succinate dehydrogenase and MYC-associated factor X mutations in pituitary neuroendocrine tumours
Abstract
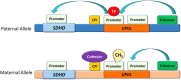
Pituitary neuroendocrine tumours (PitNETs) associated with paragangliomas or phaeochromocytomas are rare. SDHx variants are estimated to be associated with 0.3-1.8% of PitNETs. Only a few case reports have documented the association with MAX variants. Prolactinomas are the most common PitNETs occurring in patients with SDHx variants, followed by somatotrophinomas, clinically non-functioning tumours and corticotrophinomas. One pituitary carcinoma has been described. SDHC, SDHB and SDHA mutations are inherited in an autosomal dominant fashion and tumorigenesis seems to adhere to Knudson's two-hit hypothesis. SDHD and SDHAF2 mutations most commonly have paternal inheritance. Immunohistochemistry for SDHB or MAX and loss of heterozygosity analysis can support the assessment of pathogenicity of the variants. Metabolomics is promising in the diagnosis of SDHx-related disease. Future research should aim to further clarify the role of SDHx and MAX variants or other genes in the molecular pathogenesis of PitNETs, including pseudohypoxic and kinase signalling pathways along with elucidating epigenetic mechanisms to predict tumour behaviour.
Keywords: MAX; SDH; paraganglioma; phaeochromocytoma; pituitary neuroendocrine tumour; succinate dehydrogenase.
Figures
Similar articles
-
Non-pheochromocytoma (PCC)/paraganglioma (PGL) tumors in patients with succinate dehydrogenase-related PCC-PGL syndromes: a clinicopathological and molecular analysis.Eur J Endocrinol. 2013 Nov 22;170(1):1-12. doi: 10.1530/EJE-13-0623. Print 2014 Jan. Eur J Endocrinol. 2013. PMID: 24096523
-
Potential Pitfalls of SDH Immunohistochemical Detection in Paragangliomas and Phaeochromocytomas Harbouring Germline SDHx Gene Mutation.Anticancer Res. 2017 Feb;37(2):805-812. doi: 10.21873/anticanres.11381. Anticancer Res. 2017. PMID: 28179334
-
Succinate Dehydrogenase (SDH)-Deficient Pancreatic Neuroendocrine Tumor Expands the SDH-Related Tumor Spectrum.J Clin Endocrinol Metab. 2015 Oct;100(10):E1386-93. doi: 10.1210/jc.2015-2689. Epub 2015 Aug 10. J Clin Endocrinol Metab. 2015. PMID: 26259135
-
A review of the tumour spectrum of germline succinate dehydrogenase gene mutations: Beyond phaeochromocytoma and paraganglioma.Clin Endocrinol (Oxf). 2020 Nov;93(5):528-538. doi: 10.1111/cen.14289. Epub 2020 Aug 14. Clin Endocrinol (Oxf). 2020. PMID: 32686200 Review.
-
Risk of metastatic pheochromocytoma and paraganglioma in SDHx mutation carriers: a systematic review and updated meta-analysis.J Med Genet. 2020 Apr;57(4):217-225. doi: 10.1136/jmedgenet-2019-106324. Epub 2019 Oct 24. J Med Genet. 2020. PMID: 31649053
Cited by
-
SDHx mutation and pituitary adenoma: can in vivo 1H-MR spectroscopy unravel the link?Endocr Relat Cancer. 2023 Jan 5;30(2):e220198. doi: 10.1530/ERC-22-0198. Print 2023 Feb 1. Endocr Relat Cancer. 2023. PMID: 36449569 Free PMC article.
-
Childhood Multiple Endocrine Neoplasia (MEN) Syndromes: Genetics, Clinical Heterogeneity and Modifying Genes.J Clin Med. 2024 Sep 18;13(18):5510. doi: 10.3390/jcm13185510. J Clin Med. 2024. PMID: 39336996 Free PMC article. Review.
-
MAX inactivation deregulates the MYC network and induces neuroendocrine neoplasia in multiple tissues.bioRxiv [Preprint]. 2024 Sep 24:2024.09.21.614255. doi: 10.1101/2024.09.21.614255. bioRxiv. 2024. Update in: Sci Adv. 2025 Apr 25;11(17):eadt3177. doi: 10.1126/sciadv.adt3177. PMID: 39386474 Free PMC article. Updated. Preprint.
-
MAX inactivation deregulates the MYC network and induces neuroendocrine neoplasia in multiple tissues.Sci Adv. 2025 Apr 25;11(17):eadt3177. doi: 10.1126/sciadv.adt3177. Epub 2025 Apr 25. Sci Adv. 2025. PMID: 40279415 Free PMC article.
-
A rare association of pheochromocytoma, paraganglioma, and pituitary adenoma (3PA): A case report and literature review.Medicine (Baltimore). 2024 Aug 9;103(32):e38928. doi: 10.1097/MD.0000000000038928. Medicine (Baltimore). 2024. PMID: 39121253 Free PMC article. Review.
References
-
- Andrews KA, Ascher DB, Pires DEV, Barnes DR, Vialard L, Casey RT, Bradshaw N, Adlard J, Aylwin S, Brennan Pet al.2018Tumour risks and genotype-phenotype correlations associated with germline variants in succinate dehydrogenase subunit genes SDHB, SDHC and SDHD. Journal of Medical Genetics 55384–394. (10.1136/jmedgenet-2017-105127) - DOI - PMC - PubMed
-
- Benn DE, Gimenez-Roqueplo AP, Reilly JR, Bertherat J, Burgess J, Byth K, Croxson M, Dahia PL, Elston M, Gimm O, et al. 2006Clinical presentation and penetrance of pheochromocytoma/paraganglioma syndromes. Journal of Clinical Endocrinology and Metabolism 91 827–836. (10.1210/jc.2005-1862) - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

