The structure-specific endonuclease complex SLX4-XPF regulates Tus-Ter-induced homologous recombination
- PMID: 35941380
- PMCID: PMC9941964
- DOI: 10.1038/s41594-022-00812-9
The structure-specific endonuclease complex SLX4-XPF regulates Tus-Ter-induced homologous recombination
Abstract
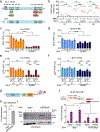
Vertebrate replication forks arrested at interstrand DNA cross-links (ICLs) engage the Fanconi anemia pathway to incise arrested forks, 'unhooking' the ICL and forming a double strand break (DSB) that is repaired by homologous recombination (HR). The FANCP product, SLX4, in complex with the XPF (also known as FANCQ or ERCC4)-ERCC1 endonuclease, mediates ICL unhooking. Whether this mechanism operates at replication fork barriers other than ICLs is unknown. Here, we study the role of mouse SLX4 in HR triggered by a site-specific chromosomal DNA-protein replication fork barrier formed by the Escherichia coli-derived Tus-Ter complex. We show that SLX4-XPF is required for Tus-Ter-induced HR but not for error-free HR induced by a replication-independent DSB. We additionally uncover a role for SLX4-XPF in DSB-induced long-tract gene conversion, an error-prone HR pathway related to break-induced replication. Notably, Slx4 and Xpf mutants that are defective for Tus-Ter-induced HR are hypersensitive to ICLs and also to the DNA-protein cross-linking agents 5-aza-2'-deoxycytidine and zebularine. Collectively, these findings show that SLX4-XPF can process DNA-protein fork barriers for HR and that the Tus-Ter system recapitulates this process.
© 2022. The Author(s), under exclusive licence to Springer Nature America, Inc.
Conflict of interest statement
Declaration of Interests
The authors declare no competing interests.
Figures














References
-
- Berti M, Cortez D. & Lopes M. The plasticity of DNA replication forks in response to clinically relevant genotoxic stress. Nat Rev Mol Cell Biol 21, 633–651 (2020). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

