Role of Mass Transport in the Deposition, Growth, and Transformation of Calcium Carbonate on Surfaces at High Supersaturation
- PMID: 35942121
- PMCID: PMC9354018
- DOI: 10.1021/acs.cgd.1c01505
Role of Mass Transport in the Deposition, Growth, and Transformation of Calcium Carbonate on Surfaces at High Supersaturation
Abstract
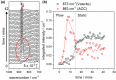
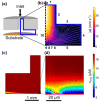
We demonstrate how combined in-situ measurements and finite element method modeling can provide new insight into the relative contribution of mass transport to the growth of calcium carbonate on two model surfaces, glass and gold, under high-supersaturation conditions relevant to surface scaling. An impinging jet-radial flow system is used to create a high-supersaturated solution at the inlet of different cells: an optical microscope cell presenting a glass surface for deposition and quartz crystal microbalance (QCM) and in-situ IR spectroscopy cells, both presenting a gold surface. The approach described is quantitative due to the well-defined mass transport, and both time-lapse optical microscopy images and QCM data are analyzed to provide information on the growth kinetics of the calcite crystals. Initially, amorphous calcium carbonate (ACC), formed in solution, dominates the deposition process. At longer times, the growth of calcite is more significant and, on glass, is observed to consume ACC from the surface, leading to surface regions depleted of ACC developing around calcite microcrystals. On Au, the mass increase becomes linear with time in this region. Taken together, these microscopic and macroscopic measurements demonstrate that calcite growth has a significant component of mass transport control at high supersaturation. Finite element method (FEM) simulations of mass-transport-limited crystal growth support the strong mass transport contribution to the growth kinetics and further suggest that the observed growth must be sustained by more than just the Ca2+ and CO3 2- in solution, with dissolution/direct attachment of ACC and/or ion pairs also contributing to the growth process.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures







References
-
- Dalas E.; Koutsoukos P. G. Calcium Carbonate Scale Formation and Prevention in a Flow-through System at Various Temperatures. Desalination 1990, 78 (3), 403–416. 10.1016/0011-9164(90)80060-O. - DOI
-
- Falini G.; Albeck S.; Weiner S.; Addadi L. Control of Aragonite or Calcite Polymorphism by Mollusk Shell Macromolecules. Science 1996, 271 (5245), 67–69. 10.1126/science.271.5245.67. - DOI
-
- de Yoreo J. J.; Waychunas G. A.; Jun Y.-S.; Fernandez-Martinez A.. 7. In Situ Investigations of Carbonate Nucleation on Mineral and Organic Surfaces. In Reviews in Mineralogy & Geochemistry; De Gruyter, 2013; Vol. 77, pp 229–258. 10.1515/9781501508073-009. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous
