Effect of Stenting Plus Medical Therapy vs Medical Therapy Alone on Risk of Stroke and Death in Patients With Symptomatic Intracranial Stenosis: The CASSISS Randomized Clinical Trial
- PMID: 35943472
- PMCID: PMC9364128
- DOI: 10.1001/jama.2022.12000
Effect of Stenting Plus Medical Therapy vs Medical Therapy Alone on Risk of Stroke and Death in Patients With Symptomatic Intracranial Stenosis: The CASSISS Randomized Clinical Trial
Abstract
Importance: Prior randomized trials have generally shown harm or no benefit of stenting added to medical therapy for patients with symptomatic severe intracranial atherosclerotic stenosis, but it remains uncertain as to whether refined patient selection and more experienced surgeons might result in improved outcomes.
Objective: To compare stenting plus medical therapy vs medical therapy alone in patients with symptomatic severe intracranial atherosclerotic stenosis.
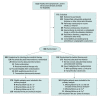
Design, setting, and participants: Multicenter, open-label, randomized, outcome assessor-blinded trial conducted at 8 centers in China. A total of 380 patients with transient ischemic attack or nondisabling, nonperforator (defined as nonbrainstem or non-basal ganglia end artery) territory ischemic stroke attributed to severe intracranial stenosis (70%-99%) and beyond a duration of 3 weeks from the latest ischemic symptom onset were recruited between March 5, 2014, and November 10, 2016, and followed up for 3 years (final follow-up: November 10, 2019).
Interventions: Medical therapy plus stenting (n = 176) or medical therapy alone (n = 182). Medical therapy included dual-antiplatelet therapy for 90 days (single antiplatelet therapy thereafter) and stroke risk factor control.
Main outcomes and measures: The primary outcome was a composite of stroke or death within 30 days or stroke in the qualifying artery territory beyond 30 days through 1 year. There were 5 secondary outcomes, including stroke in the qualifying artery territory at 2 years and 3 years as well as mortality at 3 years.
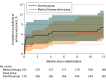
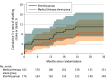
Results: Among 380 patients who were randomized, 358 were confirmed eligible (mean age, 56.3 years; 263 male [73.5%]) and 343 (95.8%) completed the trial. For the stenting plus medical therapy group vs medical therapy alone, no significant difference was found for the primary outcome of risk of stroke or death (8.0% [14/176] vs 7.2% [13/181]; difference, 0.4% [95% CI, -5.0% to 5.9%]; hazard ratio, 1.10 [95% CI, 0.52-2.35]; P = .82). Of the 5 prespecified secondary end points, none showed a significant difference including stroke in the qualifying artery territory at 2 years (9.9% [17/171] vs 9.0% [16/178]; difference, 0.7% [95% CI, -5.4% to 6.7%]; hazard ratio, 1.10 [95% CI, 0.56-2.16]; P = .80) and 3 years (11.3% [19/168] vs 11.2% [19/170]; difference, -0.2% [95% CI, -7.0% to 6.5%]; hazard ratio, 1.00 [95% CI, 0.53-1.90]; P > .99). Mortality at 3 years was 4.4% (7/160) in the stenting plus medical therapy group vs 1.3% (2/159) in the medical therapy alone group (difference, 3.2% [95% CI, -0.5% to 6.9%]; hazard ratio, 3.75 [95% CI, 0.77-18.13]; P = .08).
Conclusions and relevance: Among patients with transient ischemic attack or ischemic stroke due to symptomatic severe intracranial atherosclerotic stenosis, the addition of percutaneous transluminal angioplasty and stenting to medical therapy, compared with medical therapy alone, resulted in no significant difference in the risk of stroke or death within 30 days or stroke in the qualifying artery territory beyond 30 days through 1 year. The findings do not support the addition of percutaneous transluminal angioplasty and stenting to medical therapy for the treatment of patients with symptomatic severe intracranial atherosclerotic stenosis.
Trial registration: ClinicalTrials.gov Identifier: NCT01763320.
Conflict of interest statement
Figures
Comment in
-
Stenting Plus Medical Therapy and Risk of Stroke and Death in Patients With Symptomatic Intracranial Stenosis.JAMA. 2022 Dec 27;328(24):2456-2457. doi: 10.1001/jama.2022.18918. JAMA. 2022. PMID: 36573984 No abstract available.
-
Stenting Plus Medical Therapy and Risk of Stroke and Death in Patients With Symptomatic Intracranial Stenosis.JAMA. 2022 Dec 27;328(24):2455-2456. doi: 10.1001/jama.2022.18915. JAMA. 2022. PMID: 36573985 No abstract available.

