Tigecycline Soft Tissue Penetration in Obese and Non-obese Surgical Patients Determined by Using In Vivo Microdialysis
- PMID: 35945479
- PMCID: PMC9399032
- DOI: 10.1007/s13318-022-00789-2
Tigecycline Soft Tissue Penetration in Obese and Non-obese Surgical Patients Determined by Using In Vivo Microdialysis
Abstract
Background and objective: Tigecycline, a broad-spectrum glycylcycline antibiotic, is approved for use at a fixed dose irrespective of body weight. However, its pharmacokinetics may be altered in obesity, which would impact on the antibiotic's effectiveness. The objective of this study was to investigate the plasma and subcutaneous tissue concentrations of tigecycline in obese patients compared with those in a non-obese control group.
Methods: Fifteen obese patients (one class II and 14 class III) undergoing bariatric surgery and 15 non-obese patients undergoing intra-abdominal surgery (mainly tumour resection) received a single dose of 50 or 100 mg tigecycline as an intravenous short infusion. Tigecycline concentrations were measured up to 8 h after dosing in plasma (total concentration), in ultrafiltrate of plasma (free concentration), and in microdialysate from subcutaneous tissue, respectively.
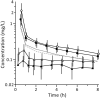
Results: In obese patients, total peak plasma concentration (1.31 ± 0.50 vs 2.27 ± 1.40 mg/L) and the area under the concentration-time curve from 0 to 8 h (AUC8h,plasma: 2.15 ± 0.42 vs 2.74 ± 0.73 h⋅mg/L), as normalized to a 100 mg dose, were significantly lower compared with those of non-obese patients. No significant differences were observed regarding the free plasma concentration, as determined by ultrafiltration, or the corresponding AUC8h (fAUC8h,plasma). Concentrations in interstitial fluid (ISF) of subcutaneous tissue were lower than the free plasma concentrations in both groups, and they were lower in obese compared to non-obese patients: the AUC8h in ISF (AUC8h,ISF) was 0.51 ± 0.22 h⋅mg/L in obese and 0.79 ± 0.23 h⋅mg/L in non-obese patients, resulting in a relative tissue drug exposure (AUC8h,ISF/fAUC8h,plasma) of 0.38 ± 0.19 and 0.63 ± 0.24, respectively.
Conclusion: Following a single dose of tigecycline, concentrations in the ISF of subcutaneous adipose tissue are decreased in heavily obese subjects, calling for an increased loading dose.
Eu clinical trials registration number: EudraCT No. 2012-004383-22.
© 2022. The Author(s).
Conflict of interest statement
CK reports research grants for PharMetrX, DDMoRe, FAIR. HW received grants from Pfizer (Investigator Initiated Trial Program, Berlin, Germany) and InfectoPharm (Heppenheim, Germany), both for the clinical microdialysis trial. HW reports lecture fees from InfectoPharm (Heppenheim, Germany), MSD (Konstanz, Germany), Getinge (Rastatt, Germany) and Medtronic (Meerbusch, Germany) and consultant honoraria from Dräger Medical (Lübeck, Germany) and Liberate Medical (Crestwood, KY, USA). PS reports lecture fees from InfectoPharm (Heppenheim, Germany). The other authors have no conflicts of interest to declare.
Figures
Similar articles
-
Soft tissue penetration of cefuroxime determined by clinical microdialysis in morbidly obese patients undergoing abdominal surgery.Int J Antimicrob Agents. 2009 Sep;34(3):231-5. doi: 10.1016/j.ijantimicag.2009.03.019. Epub 2009 May 2. Int J Antimicrob Agents. 2009. PMID: 19411164 Clinical Trial.
-
Comparative Plasma and Interstitial Tissue Fluid Pharmacokinetics of Meropenem Demonstrate the Need for Increasing Dose and Infusion Duration in Obese and Non-obese Patients.Clin Pharmacokinet. 2022 May;61(5):655-672. doi: 10.1007/s40262-021-01070-6. Epub 2021 Dec 11. Clin Pharmacokinet. 2022. PMID: 34894344 Free PMC article. Clinical Trial.
-
Meropenem Plasma and Interstitial Soft Tissue Concentrations in Obese and Nonobese Patients-A Controlled Clinical Trial.Antibiotics (Basel). 2020 Dec 21;9(12):931. doi: 10.3390/antibiotics9120931. Antibiotics (Basel). 2020. PMID: 33371322 Free PMC article.
-
A comprehensive review on the pharmacokinetics of antibiotics in interstitial fluid spaces in humans: implications on dosing and clinical pharmacokinetic monitoring.Clin Pharmacokinet. 2014 Aug;53(8):695-730. doi: 10.1007/s40262-014-0152-3. Clin Pharmacokinet. 2014. PMID: 24972859 Review.
-
Tigecycline: a glycylcycline antimicrobial agent.Clin Ther. 2006 Aug;28(8):1079-1106. doi: 10.1016/j.clinthera.2006.08.011. Clin Ther. 2006. PMID: 16982286 Review.
Cited by
-
Penetration of Antibiotics into Subcutaneous and Intramuscular Interstitial Fluid: A Meta-Analysis of Microdialysis Studies in Adults.Clin Pharmacokinet. 2024 Jul;63(7):965-980. doi: 10.1007/s40262-024-01394-z. Epub 2024 Jul 2. Clin Pharmacokinet. 2024. PMID: 38955946
-
Dynamic distribution of systemically administered antibiotics in orthopeadically relevant target tissues and settings.APMIS. 2024 Dec;132(12):992-1025. doi: 10.1111/apm.13490. Epub 2024 Nov 12. APMIS. 2024. PMID: 39530161 Free PMC article.
-
Updated antimicrobial dosing recommendations for obese patients.Antimicrob Agents Chemother. 2024 May 2;68(5):e0171923. doi: 10.1128/aac.01719-23. Epub 2024 Mar 25. Antimicrob Agents Chemother. 2024. PMID: 38526051 Free PMC article.
-
Machine learning-based prediction model for hypofibrinogenemia after tigecycline therapy.BMC Med Inform Decis Mak. 2024 Oct 4;24(1):284. doi: 10.1186/s12911-024-02694-x. BMC Med Inform Decis Mak. 2024. PMID: 39367370 Free PMC article.
References
-
- Wyeth Pharmaceuticals LLC. TYGACIL® (tigecycline) for injection, for intravenous use, Prescribing information [Internet]. 2021. Available from: https://labeling.pfizer.com/ShowLabeling.aspx?id=491.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

