Diagnostic and prognostic nomograms for newly diagnosed intrahepatic cholangiocarcinoma with brain metastasis: A population-based analysis
- PMID: 35946168
- PMCID: PMC9597213
- DOI: 10.1177/15353702221113828
Diagnostic and prognostic nomograms for newly diagnosed intrahepatic cholangiocarcinoma with brain metastasis: A population-based analysis
Abstract
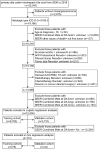
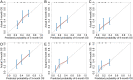
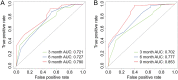
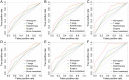
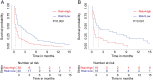
Brain metastasis (BM) is one of the rare metastatic sites of intrahepatic cholangiocarcinoma (ICC). ICC with BM can seriously affect the quality of life of patients and lead to a poor prognosis. The aim of this study was to establish two nomograms to estimate the risk of BM in ICC patients and the prognosis of ICC patients with BM. Data on 19,166 individuals diagnosed with ICC were retrospectively collected from the Surveillance, Epidemiology, and End Results (SEER) database. Independent risk factors and prognostic factors were identified by the logistic and the Cox regression, respectively. Next, two nomograms were developed, and their discrimination was estimated by concordance index (C-index) and calibration plots, while the clinical benefits of the prognostic nomogram were evaluated using the receiver operating characteristic (ROC) curves, the decision curve analysis (DCA), and the Kaplan-Meier analyses. The independent risk factors for BM were T stage, N stage, surgery, alpha-fetoprotein (AFP) level, and tumor size. T stage, surgery, radiotherapy, and bone metastasis were prognostic factors for overall survival (OS). For the prognostic nomogram, the C-index was 0.759 (95% confidence interval (CI) = 0.745-0.773) and 0.764 (95% CI = 0.747-0.781) in the training and the validation cohort, respectively. The calibration curves revealed a robust agreement between predictions and actual observations probability. The area under curves (AUCs) for the 3-, 6-, and 9-month OS were 0.721, 0.727, and 0.790 in the training cohort and 0.702, 0.777, and 0.853 in the validation cohort, respectively. The DCA curves yielded remarkable positive net benefits over a wide range of threshold probabilities. The Kaplan-Meier analysis illustrated that the nomogram could significantly distinguish the population with different survival risks. We successfully established the two nomograms for predicting the incidence of BM and the prognosis of ICC patients with BM, which may assist clinicians in choosing more effective treatment strategies.
Keywords: Epidemiology; Intrahepatic cholangiocarcinoma; Surveillance; and End Results; brain metastasis; diagnosis; nomogram; prognosis.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures




Similar articles
-
Development and validation of a diagnostic and prognostic model for bone metastasis of intrahepatic cholangiocarcinoma: a population-based analysis.Transl Cancer Res. 2024 Aug 31;13(8):4010-4027. doi: 10.21037/tcr-24-567. Epub 2024 Aug 27. Transl Cancer Res. 2024. PMID: 39262477 Free PMC article.
-
The prognostic value of sarcopenia combined with preoperative fibrinogen-albumin ratio in patients with intrahepatic cholangiocarcinoma after surgery: A multicenter, prospective study.Cancer Med. 2021 Jul;10(14):4768-4780. doi: 10.1002/cam4.4035. Epub 2021 Jun 8. Cancer Med. 2021. PMID: 34105304 Free PMC article.
-
Predictors of Distant Metastasis and Prognosis in Newly Diagnosed T1 Intrahepatic Cholangiocarcinoma.Biomed Res Int. 2023 Jan 17;2023:6638755. doi: 10.1155/2023/6638755. eCollection 2023. Biomed Res Int. 2023. PMID: 36704724 Free PMC article. Review.
-
Establishment and external validation of prognosis prediction nomogram for patients with distant metastatic intrahepatic cholangiocarcinoma: based on a large population.BMC Cancer. 2024 Feb 16;24(1):227. doi: 10.1186/s12885-024-11976-6. BMC Cancer. 2024. PMID: 38365630 Free PMC article.
-
Development and validation of web-based nomograms for predicting survival status in patients with intrahepatic cholangiocarcinoma depending on the surgical status: a SEER database analysis.Sci Rep. 2024 Jan 18;14(1):1568. doi: 10.1038/s41598-024-52025-3. Sci Rep. 2024. PMID: 38238494 Free PMC article.
Cited by
-
An explainable machine learning model for predicting the risk of distant metastasis in intrahepatic cholangiocarcinoma: a population-based cohort study.Discov Oncol. 2025 Jun 18;16(1):1140. doi: 10.1007/s12672-025-02952-y. Discov Oncol. 2025. PMID: 40531423 Free PMC article.
-
Pemigatinib combined with immunotherapy and stereotactic body radiation therapy for FGFR2 fusion-positive advanced intrahepatic cholangiocarcinoma with brain metastasis: a Case Report.Front Pharmacol. 2024 Dec 4;15:1509891. doi: 10.3389/fphar.2024.1509891. eCollection 2024. Front Pharmacol. 2024. PMID: 39697545 Free PMC article.
-
Development and validation of a diagnostic and prognostic model for bone metastasis of intrahepatic cholangiocarcinoma: a population-based analysis.Transl Cancer Res. 2024 Aug 31;13(8):4010-4027. doi: 10.21037/tcr-24-567. Epub 2024 Aug 27. Transl Cancer Res. 2024. PMID: 39262477 Free PMC article.
-
Predictive models and treatment efficacy for liver cancer patients with bone metastases: A comprehensive analysis of prognostic factors and nomogram development.Heliyon. 2024 Sep 19;10(19):e38038. doi: 10.1016/j.heliyon.2024.e38038. eCollection 2024 Oct 15. Heliyon. 2024. PMID: 39386874 Free PMC article.
References
-
- Banales JM, Marin JJG, Lamarca A, Rodrigues PM, Khan SA, Roberts LR, Cardinale V, Carpino G, Andersen JB, Braconi C, Calvisi DF, Perugorria MJ, Fabris L, Boulter L, Macias RIR, Gaudio E, Alvaro D, Gradilone SA, Strazzabosco M, Marzioni M, Coulouarn C, Fouassier L, Raggi C, Invernizzi P, Mertens JC, Moncsek A, Rizvi S, Heimbach J, Koerkamp BG, Bruix J, Forner A, Bridgewater J, Valle JW, Gores GJ. Cholangiocarcinoma 2020: the next horizon in mechanisms and management. Nat Rev Gastroenterol Hepatol 2020;17:557–88 - PMC - PubMed
-
- Zhang H, Yang T, Wu M, Shen F. Intrahepatic cholangiocarcinoma: epidemiology, risk factors, diagnosis and surgical management. Cancer Lett 2016;379:198–205 - PubMed
-
- Lee YT, Wang JJ, Luu M, Noureddin M, Nissen NN, Patel TC, Roberts LR, Singal AG, Gores GJ, Yang JD. Comparison of clinical features and outcomes between intrahepatic cholangiocarcinoma and hepatocellular carcinoma in the United States. Hepatology 2021;74:2622–32 - PubMed
-
- Spolverato G, Kim Y, Ejaz A, Alexandrescu S, Marques H, Aldrighetti L, Gamblin TC, Pulitano C, Bauer TW, Shen F, Sandroussi C, Poultsides G, Maithel SK, Pawlik TM. Conditional probability of long-term survival after liver resection for intrahepatic cholangiocarcinoma: a multi-institutional analysis of 535 patients. JAMA Surg 2015;150:538–45 - PubMed
-
- Mazzaferro V, Gorgen A, Roayaie S, Droz Dit Busset M, Sapisochin G. Liver resection and transplantation for intrahepatic cholangiocarcinoma. J Hepatol 2020;72:364–77 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

