A recent update on new synthetic chiral compounds with antileishmanial activity
- PMID: 35947400
- PMCID: PMC9543214
- DOI: 10.1002/chir.23494
A recent update on new synthetic chiral compounds with antileishmanial activity
Abstract
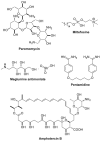
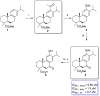
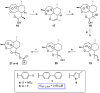
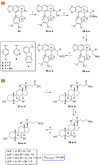
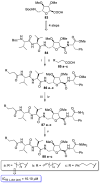
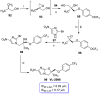
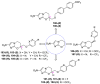
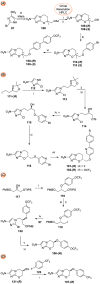
Parasitic diseases, including malaria, leishmaniasis, and trypanosomiasis, affect billions of people and are responsible for almost 500,000 deaths/year. In particular, leishmaniasis, a neglected tropical disease, is considered a global public health problem because current drugs have several drawbacks including to toxicity, high cost, and drug resistance, which result in a lack of effective and readily available therapies. Therefore, the synthesis of new, safe, and effective molecules still requires the attention of the scientific community. Moreover, it is well known that chirality plays a crucial role in the antiparasitic activity of molecules, driving the design of their synthesis. Therefore, in this review we report a recent update on new chiral compounds with promising antileishmanial activity, focusing on synthetic approaches. Where reported, in most cases the enantiopure compound has shown better potency against the protozoa than its enantiomer or corresponding racemic mixture.
Keywords: chiral catalysis; chiral pool; chiral resolution; imidazoxazines; imidazoxazoles; kinetic resolution; leishmania; medicinal chemistry; natural products; peptides.
© 2022 The Authors. Chirality published by Wiley Periodicals LLC.
Figures

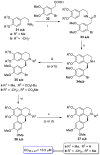
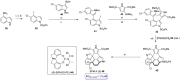
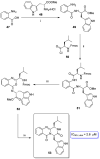
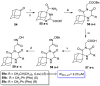
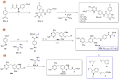
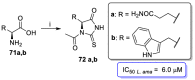
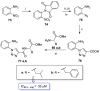
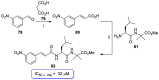

References
-
- World Health Organization Website about Leishmaniasis: www.who.int/news-room/fact-sheets/detail/leishmaniasis (accessed on 9 May 2022).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

