Single-injection COVID-19 subunit vaccine elicits potent immune responses
- PMID: 35948176
- PMCID: PMC9357281
- DOI: 10.1016/j.actbio.2022.08.006
Single-injection COVID-19 subunit vaccine elicits potent immune responses
Abstract
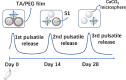
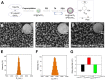
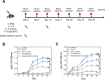
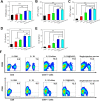
Current vaccination schedules, including COVID-19 vaccines, require multiple doses to be administered. Single injection vaccines eliciting equivalent immune response are highly desirable. Unfortunately because unconventional release kinetics are difficult to achieve it still remains a huge challenge. Herein a single-injection COVID-19 vaccine was designed using a highly programmable release system based on dynamic layer-by-layer (LBL) films. The antigen, S1 subunit of SARS-CoV-2 spike protein, was loaded in CaCO3 microspheres, which were further coated with tannic acid (TA)/polyethylene glycol (PEG) LBL films. The single-injection vaccine was obtained by mixing the microspheres coated with different thickness of TA/PEG films. Because of the unique constant-rate erosion behavior of the TA/PEG coatings, this system allows for distinct multiple pulsatile release of antigen, closely mimicking the release profile of antigen in conventional multiple dose vaccines. Immunization with the single injection vaccine induces potent and persistent S1-specific humoral and cellular immune responses in mice. The sera from the vaccinated animal exhibit robust in vitro viral neutralization ability. More importantly, the immune response and viral inhibition induced by the single injection vaccine are as strong as that induced by the corresponding multiple dose vaccine, because they share the same antigen release profile. STATEMENT OF SIGNIFICANCE: Vaccines are the most powerful and cost-effective weapons against infectious diseases such as COVID-19. However, current vaccination schedules, including the COVID-19 vaccines, require multiple doses to be administered. Herein a single-injection COVID-19 vaccine is designed using a highly programmable release system. This vaccine releases antigens in a pulsatile manner, closely mimicking the release pattern of antigens in conventional multiple dose vaccines. As a result, one single injection of the new vaccine induces an immune response and viral inhibition similar to that induced by the corresponding multiple-dose vaccine approach.
Keywords: COVID-19 vaccines; Pulsatile release; Single-injection vaccines; Unconventional release kinetics.
Copyright © 2022. Published by Elsevier Ltd.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





References
-
- C.R.C. Johns Hopkins University, COVID-19 dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). https://coronavirus.jhu.edu/map.html (accessed march 7, 2022).
-
- Shin M.D., Shukla S., Chung Y.H., Beiss V., Chan S.K., Ortega-Rivera O.A., Wirth D.M., Chen A., Sack M., Pokorski J.K., Steinmetz N.F. COVID-19 vaccine development and a potential nanomaterial path forward. Nat. Nanotechnol. 2020;15(8):646–655. - PubMed
-
- Liang J.G., Su D., Song T., Zeng Y., Huang W., Wu J., Xu R., Luo P., Yang X., Zhang X., Luo S., Liang Y., Li X., Huang J., Wang Q., Huang X., Xu Q., Luo M., Huang A., Luo D., Zhao C., Yang F., Han J., Zheng Y., Liang P. S-Trimer, a COVID-19 subunit vaccine candidate, induces protective immunity in nonhuman primates. Nat. Commun. 2021;12(1):1346. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

