Conserved subcortical processing in visuo-vestibular gaze control
- PMID: 35948549
- PMCID: PMC9365791
- DOI: 10.1038/s41467-022-32379-w
Conserved subcortical processing in visuo-vestibular gaze control
Abstract
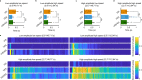
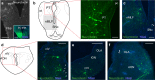
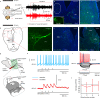
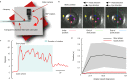
Gaze stabilization compensates for movements of the head or external environment to minimize image blurring. Multisensory information stabilizes the scene on the retina via the vestibulo-ocular (VOR) and optokinetic (OKR) reflexes. While the organization of neuronal circuits underlying VOR is well-described across vertebrates, less is known about the contribution and evolution of the OKR and the basic structures allowing visuo-vestibular integration. To analyze these neuronal pathways underlying visuo-vestibular integration, we developed a setup using a lamprey eye-brain-labyrinth preparation, which allowed coordinating electrophysiological recordings, vestibular stimulation with a moving platform, and visual stimulation via screens. Lampreys exhibit robust visuo-vestibular integration, with optokinetic information processed in the pretectum that can be downregulated from tectum. Visual and vestibular inputs are integrated at several subcortical levels. Additionally, saccades are present in the form of nystagmus. Thus, all basic components of the visuo-vestibular control of gaze were present already at the dawn of vertebrate evolution.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
Long term visuo-vestibular mismatch in freely behaving mice differentially affects gaze stabilizing reflexes.Sci Rep. 2020 Nov 18;10(1):20018. doi: 10.1038/s41598-020-77026-w. Sci Rep. 2020. PMID: 33208812 Free PMC article.
-
Senescence of human visual-vestibular interactions: smooth pursuit, optokinetic, and vestibular control of eye movements with aging.Exp Brain Res. 1994;98(2):355-72. doi: 10.1007/BF00228423. Exp Brain Res. 1994. PMID: 8050519
-
Adaptation of vestibulo-ocular and optokinetic reflexes after massed and spaced vestibulo-ocular motor learning.Behav Brain Res. 2022 May 24;426:113837. doi: 10.1016/j.bbr.2022.113837. Epub 2022 Mar 11. Behav Brain Res. 2022. PMID: 35288176
-
Plasticity in the vestibulo-ocular and optokinetic reflexes following modification of canal input.Rev Oculomot Res. 1985;1:145-53. Rev Oculomot Res. 1985. PMID: 3940026 Review.
-
Visual-vestibular interaction in the control of head and eye movement: the role of visual feedback and predictive mechanisms.Prog Neurobiol. 1993 Oct;41(4):435-72. doi: 10.1016/0301-0082(93)90026-o. Prog Neurobiol. 1993. PMID: 8210413 Review. No abstract available.
Cited by
-
Neural mechanisms to incorporate visual counterevidence in self-movement estimation.Curr Biol. 2023 Nov 20;33(22):4960-4979.e7. doi: 10.1016/j.cub.2023.10.011. Epub 2023 Nov 1. Curr Biol. 2023. PMID: 37918398 Free PMC article.
-
Unravelling the functional development of vertebrate pathways controlling gaze.Front Cell Dev Biol. 2023 Oct 26;11:1298486. doi: 10.3389/fcell.2023.1298486. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37965576 Free PMC article.
-
Properties of Gaze Strategies Based on Eye-Head Coordination in a Ball-Catching Task.Vision (Basel). 2024 Apr 15;8(2):20. doi: 10.3390/vision8020020. Vision (Basel). 2024. PMID: 38651441 Free PMC article.
-
A Proposed Mechanism for Visual Vertigo: Post-Concussion Patients Have Higher Gain From Visual Input Into Subcortical Gaze Stabilization.Invest Ophthalmol Vis Sci. 2024 Apr 1;65(4):26. doi: 10.1167/iovs.65.4.26. Invest Ophthalmol Vis Sci. 2024. PMID: 38607620 Free PMC article.
-
Roles for cerebellum and subsumption architecture in central pattern generation.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2024 Mar;210(2):315-324. doi: 10.1007/s00359-023-01634-w. Epub 2023 May 2. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2024. PMID: 37130955 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

