Synthesis of vacant graphitic carbon nitride in argon atmosphere and its utilization for photocatalytic hydrogen generation
- PMID: 35948580
- PMCID: PMC9365785
- DOI: 10.1038/s41598-022-17940-3
Synthesis of vacant graphitic carbon nitride in argon atmosphere and its utilization for photocatalytic hydrogen generation
Abstract
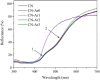
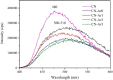
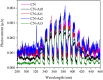
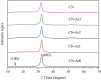
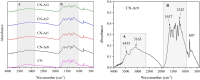
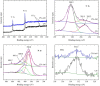
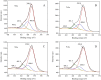
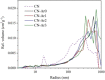
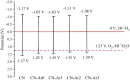
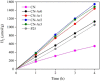
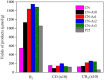
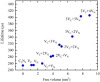
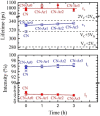
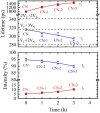
Graphitic carbon nitride (C3N4) was synthesised from melamine at 550 °C for 4 h in the argon atmosphere and then was reheated for 1-3 h at 500 °C in argon. Two band gaps of 2.04 eV and 2.47 eV were observed in all the synthetized materials. Based on the results of elemental and photoluminescence analyses, the lower band gap was found to be caused by the formation of vacancies. Specific surface areas of the synthetized materials were 15-18 m2g-1 indicating that no thermal exfoliation occurred. The photocatalytic activity of these materials was tested for hydrogen generation. The best photocatalyst showed 3 times higher performance (1547 μmol/g) than bulk C3N4 synthetized in the air (547 μmol/g). This higher activity was explained by the presence of carbon (VC) and nitrogen (VN) vacancies grouped in their big complexes 2VC + 2VN (observed by positron annihilation spectroscopy). The effect of an inert gas on the synthesis of C3N4 was demonstrated using Graham´s law of ammonia diffusion. This study showed that the synthesis of C3N4 from nitrogen-rich precursors in the argon atmosphere led to the formation of vacancy complexes beneficial for hydrogen generation, which was not referred so far.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Enhancing Photocatalytic Activity of Graphitic Carbon Nitride by Codoping with P and C for Efficient Hydrogen Generation.ACS Appl Mater Interfaces. 2017 Jul 5;9(26):21730-21737. doi: 10.1021/acsami.7b02445. Epub 2017 Jun 21. ACS Appl Mater Interfaces. 2017. PMID: 28557418
-
Comparison of Graphitic Carbon Nitrides Synthetized from Melamine and Melamine-Cyanurate Complex: Characterization and Photocatalytic Decomposition of Ofloxacin and Ampicillin.Materials (Basel). 2021 Apr 14;14(8):1967. doi: 10.3390/ma14081967. Materials (Basel). 2021. PMID: 33919916 Free PMC article.
-
Band Engineering and Morphology Control of Oxygen-Incorporated Graphitic Carbon Nitride Porous Nanosheets for Highly Efficient Photocatalytic Hydrogen Evolution.Nanomicro Lett. 2021 Jan 4;13(1):48. doi: 10.1007/s40820-020-00571-6. Nanomicro Lett. 2021. PMID: 34138228 Free PMC article.
-
Polymeric graphitic carbon nitride (g-C3N4)-based semiconducting nanostructured materials: Synthesis methods, properties and photocatalytic applications.J Environ Manage. 2019 May 15;238:25-40. doi: 10.1016/j.jenvman.2019.02.075. Epub 2019 Mar 4. J Environ Manage. 2019. PMID: 30844543 Review.
-
Metal-Free Graphitic Carbon Nitride Photocatalyst Goes Into Two-Dimensional Time.Front Chem. 2018 Dec 10;6:551. doi: 10.3389/fchem.2018.00551. eCollection 2018. Front Chem. 2018. PMID: 30619810 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

