White matter damage as a consequence of vascular dysfunction in a spontaneous mouse model of chronic mild chronic hypoperfusion with eNOS deficiency
- PMID: 35948662
- PMCID: PMC9734049
- DOI: 10.1038/s41380-022-01701-9
White matter damage as a consequence of vascular dysfunction in a spontaneous mouse model of chronic mild chronic hypoperfusion with eNOS deficiency
Abstract
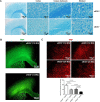
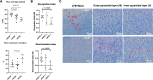
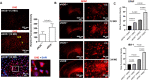
Vascular cognitive impairment and dementia (VCID) is the second most common form of dementia after Alzheimer's disease (AD). Currently, the mechanistic insights into the evolution and progression of VCID remain elusive. White matter change represents an invariant feature. Compelling clinical neuroimaging and pathological evidence suggest a link between white matter changes and neurodegeneration. Our prior study detected hypoperfused lesions in mice with partial deficiency of endothelial nitric oxide (eNOS) at very young age, precisely matching to those hypoperfused areas identified in preclinical AD patients. White matter tracts are particularly susceptible to the vascular damage induced by chronic hypoperfusion. Using immunohistochemistry, we detected severe demyelination in the middle-aged eNOS-deficient mice. The demyelinated areas were confined to cortical and subcortical areas including the corpus callosum and hippocampus. The intensity of demyelination correlated with behavioral deficits of gait and associative recognition memory performances. By Evans blue angiography, we detected blood-brain barrier (BBB) leakage as another early pathological change affecting frontal and parietal cortex in eNOS-deficient mice. Sodium nitrate fortified drinking water provided to young and middle-aged eNOS-deficient mice completely prevented non-perfusion, BBB leakage, and white matter pathology, indicating that impaired endothelium-derived NO signaling may have caused these pathological events. Furthermore, genome-wide transcriptomic analysis revealed altered gene clusters most related to mitochondrial respiratory pathways selectively in the white matter of young eNOS-deficient mice. Using eNOS-deficient mice, we identified BBB breakdown and hypoperfusion as the two earliest pathological events, resulting from insufficient vascular NO signaling. We speculate that the compromised BBB and mild chronic hypoperfusion trigger vascular damage, along with oxidative stress and astrogliosis, accounting for the white matter pathological changes in the eNOS-deficient mouse model. We conclude that eNOS-deficient mice represent an ideal spontaneous evolving model for studying the earliest events leading to white matter changes, which will be instrumental to future therapeutic testing of drug candidates and for targeting novel/specific vascular mechanisms contributing to VCID and AD.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Schneider JA, Arvanitakis Z, Bang W, Bennett DA. Mixed brain pathologies account for most dementia cases in community-dwelling older persons. Neurology. 2007;69:2197–204. - PubMed
-
- Hofman A, Ott A, Breteler MM, Bots ML, Slooter AJ, van Harskamp F, et al. Atherosclerosis, apolipoprotein E, and prevalence of dementia and Alzheimer’s disease in the Rotterdam study. Lancet. 1997;349:151–4. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

