Enhanced effect of combining bone marrow mesenchymal stem cells (BMMSCs) and pulsed electromagnetic fields (PEMF) to promote recovery after spinal cord injury in mice
- PMID: 35949547
- PMCID: PMC9350428
- DOI: 10.1002/mco2.160
Enhanced effect of combining bone marrow mesenchymal stem cells (BMMSCs) and pulsed electromagnetic fields (PEMF) to promote recovery after spinal cord injury in mice
Abstract
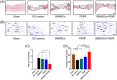
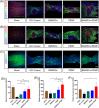
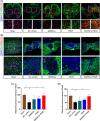
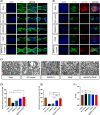
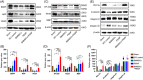
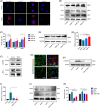
Spinal cord injury (SCI) is a destructive traumatic disease of the central nervous system without satisfying therapy efficiency. Bone marrow mesenchymal stem cells (BMMSCs) therapy promotes the neurotrophic factors' secretion and axonal regeneration, thereby promoting recovery of SCI. Pulsed electromagnetic fields (PEMF) therapy has been proven to promote neural growth and regeneration. Both BMMSCs and PEMF have shown curative effects for SCI; PEMF can further promote stem cell differentiation. Thus, we explored the combined effects of BMMSCs and PEMF and the potential interaction between these two therapies in SCI. Compared with the SCI control, BMMSCs, and PEMF groups, the combinational therapy displayed the best therapeutic effect. Combinational therapy increased the expression levels of nutritional factors including brain-derived neurotrophic factor (BDNF), nerve growth factors (NGF) and vascular endothelial growth factor (VEGF), enhanced neuron preservation (NeuN and NF-200), and increased axonal growth (MBP and myelin sheath). Additionally, PEMF promoted the expression levels of BDNF and VEGF in BMMSCs via Wnt/β-catenin signaling pathway. In summary, the combined therapy of BMMSCs and PEMF displayed a more satisfactory effect than BMMSCs and PEMF therapy alone, indicating a promising application of combined therapy for the therapy of SCI.
Keywords: bone marrow mesenchymal stem cells; motor functional recovery; nutritional factors; pulsed electromagnetic fields; spinal cord injury.
© 2022 The Authors. MedComm published by Sichuan International Medical Exchange & Promotion Association (SCIMEA) and John Wiley & Sons Australia, Ltd.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Pulsed Electromagnetic Fields Ameliorate Skeletal Deterioration in Bone Mass, Microarchitecture, and Strength by Enhancing Canonical Wnt Signaling-Mediated Bone Formation in Rats with Spinal Cord Injury.J Neurotrauma. 2021 Mar 15;38(6):765-776. doi: 10.1089/neu.2020.7296. Epub 2021 Jan 8. J Neurotrauma. 2021. PMID: 33108939
-
Effects of combination treatment with transcranial magnetic stimulation and bone marrow mesenchymal stem cell transplantation or Raf inhibition on spinal cord injury in rats.Mol Med Rep. 2021 Apr;23(4):294. doi: 10.3892/mmr.2021.11934. Epub 2021 Mar 2. Mol Med Rep. 2021. PMID: 33649786 Free PMC article.
-
Does vitamin C have the ability to augment the therapeutic effect of bone marrow-derived mesenchymal stem cells on spinal cord injury?Neural Regen Res. 2017 Dec;12(12):2050-2058. doi: 10.4103/1673-5374.221163. Neural Regen Res. 2017. PMID: 29323045 Free PMC article.
-
Cell transplantation therapies for spinal cord injury focusing on bone marrow mesenchymal stem cells: Advances and challenges.World J Stem Cells. 2023 May 26;15(5):385-399. doi: 10.4252/wjsc.v15.i5.385. World J Stem Cells. 2023. PMID: 37342219 Free PMC article. Review.
-
Bone marrow mesenchymal stem cells (BMSCs) improved functional recovery of spinal cord injury partly by promoting axonal regeneration.Neurochem Int. 2018 May;115:80-84. doi: 10.1016/j.neuint.2018.02.007. Epub 2018 Feb 16. Neurochem Int. 2018. PMID: 29458076 Review.
Cited by
-
Pulsed electromagnetic stimulation promotes neuronal maturation by up-regulating cholesterol biosynthesis.Stem Cell Res Ther. 2025 Jul 26;16(1):406. doi: 10.1186/s13287-025-04469-1. Stem Cell Res Ther. 2025. PMID: 40713855 Free PMC article.
-
Pulsed electromagnetic fields potentiate bone marrow mesenchymal stem cell chondrogenesis by regulating the Wnt/β-catenin signaling pathway.J Transl Med. 2024 Aug 6;22(1):741. doi: 10.1186/s12967-024-05470-7. J Transl Med. 2024. PMID: 39107784 Free PMC article.
-
MSCs overexpressing GDNF restores brain structure and neurological function in rats with intracerebral hemorrhage.Mol Biomed. 2023 Nov 27;4(1):43. doi: 10.1186/s43556-023-00159-7. Mol Biomed. 2023. PMID: 38008847 Free PMC article.
-
Effects of Electrical Stimulation of the Cell: Wound Healing, Cell Proliferation, Apoptosis, and Signal Transduction.Med Sci (Basel). 2023 Jan 17;11(1):11. doi: 10.3390/medsci11010011. Med Sci (Basel). 2023. PMID: 36810478 Free PMC article.
-
Wnt signaling pathway in spinal cord injury: from mechanisms to potential applications.Front Mol Neurosci. 2024 Jul 24;17:1427054. doi: 10.3389/fnmol.2024.1427054. eCollection 2024. Front Mol Neurosci. 2024. PMID: 39114641 Free PMC article. Review.
References
-
- Assinck P, Duncan GJ, Hilton BJ, Plemel JR, Tetzlaff W. Cell transplantation therapy for spinal cord injury. Nat Neurosci. 2017;20(5):637‐647. - PubMed
-
- Vismara I, Papa S, Rossi F, Forloni G, Veglianese P. Current options for cell therapy in spinal cord injury. Trends Mol Med. 2017;23(9):831‐849. - PubMed
-
- Khazaei M, Ahuja CS, Nakashima H, et al. GDNF rescues the fate of neural progenitor grafts by attenuating Notch signals in the injured spinal cord in rodents. Sci Transl Med. 2020;12(525). - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
