Therapeutic knockdown of miR-320 improves deteriorated cardiac function in a pre-clinical model of non-ischemic diabetic heart disease
- PMID: 35950211
- PMCID: PMC9356207
- DOI: 10.1016/j.omtn.2022.07.007
Therapeutic knockdown of miR-320 improves deteriorated cardiac function in a pre-clinical model of non-ischemic diabetic heart disease
Abstract
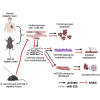
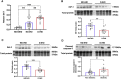
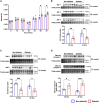
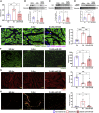
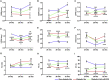
Non-ischemic diabetic heart disease (NiDHD) is characterized by diastolic dysfunction and decreased or preserved systolic function, eventually resulting in heart failure. Accelerated apoptotic cell death because of alteration of molecular signaling pathways due to dysregulation in microRNAs (miRNAs) plays a significant role in the development of NiDHD. Here, we aimed to determine the pathological role of cardiomyocyte-enriched pro-apoptotic miR-320 in the development of NiDHD. We identified a marked upregulation of miR-320 that was associated with downregulation of its target protein insulin growth factor-1 (IGF-1) in human right atrial appendage tissue in the late stages of cardiomyopathy in type 2 diabetic db/db mice and high-glucose-cultured human ventricular cardiomyocytes (AC-16 cells). In vitro knockdown of miR-320 in high-glucose-exposed AC-16 cells using locked nucleic acid (LNA) anti-miR-320 markedly reduced high-glucose-induced apoptosis by restoring IGF-1 and Bcl-2. Finally, in vivo knockdown of miR-320 in 24-week-old type 2 diabetic db/db mice reduced cardiomyocyte apoptosis and interstitial fibrosis while restoring vascular density. This resulted in partial recovery of the impaired diastolic and systolic function. Our study provides evidence that miR-320 is a late-responding miRNA that aggravates apoptosis and cardiac dysfunction in the diabetic heart, and that therapeutic knockdown of miR-320 is beneficial in partially restoring the deteriorated cardiac function.
Keywords: apoptosis; fibrosis; insulin growth factor-1; locked nucleic acid; miR-320; microRNA; microangiopathy; non-ischemic diabetic heart disease.
© 2022 The Author(s).
Conflict of interest statement
The authors declare they have no conflict of interest.
Figures


References
-
- Anderson J.L., Adams C.D., Antman E.M., Bridges C.R., Califf R.M., Casey D.E., Chavey W.E., Fesmire F.M., Hochman J.S., Levin T.N., et al. ACC/AHA 2007 guidelines for the management of patients with unstable angina/non–ST-elevation myocardial infarction: a report of the American College of Cardiology/American heart association task force on practice guidelines (writing Committee to revise the 2002 guidelines for the management of patients with unstable Angina/non–ST-elevation myocardial infarction) developed in collaboration with the American College of emergency physicians, the society for cardiovascular angiography and interventions, and the society of thoracic surgeons endorsed by the American association of cardiovascular and pulmonary rehabilitation and the society for academic emergency medicine. J. Am. Coll. Cardiol. 2007;50:e1–e157. - PubMed
-
- Bell D.S.H. Heart failure. Diabetes Care. 2003;26:2433–2441. - PubMed
-
- From A.M., Leibson C.L., Bursi F., Redfield M.M., Weston S.A., Jacobsen S.J., Rodeheffer R.J., Roger V.L. Diabetes in heart failure: prevalence and impact on outcome in the population. Am. J. Med. 2006;119:591–599. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

