α-Synuclein Interaction with Lipid Bilayer Discs
- PMID: 35952001
- PMCID: PMC9404543
- DOI: 10.1021/acs.langmuir.2c01368
α-Synuclein Interaction with Lipid Bilayer Discs
Abstract
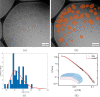
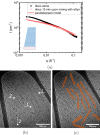
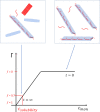
α-Synuclein (aSyn) is a 140 residue long protein present in presynaptic termini of nerve cells. The protein is associated with Parkinson's disease, in which case it has been found to self-assemble into long amyloid fibrils forming intracellular inclusions that are also rich in lipids. Furthermore, its synaptic function is proposed to involve interaction with lipid membranes, and hence, it is of interest to understand aSyn-lipid membrane interactions in detail. In this paper we report on the interaction of aSyn with model membranes in the form of lipid bilayer discs. Using a combination of cryogenic transmission electron microscopy and small-angle neutron scattering, we show that circular discs undergo a significant shape transition after the adsorption of aSyn. When aSyn self-assembles into fibrils, aSyn molecules desorb from the bilayer discs, allowing them to recover to their original shape. Interestingly, the desorption process has an all-or-none character, resulting in a binary coexistence of circular bilayer discs with no adsorbed aSyn and deformed bilayer discs having a maximum amount of adsorbed protein. The observed coexistence is consistent with the recent finding of cooperative aSyn adsorption to anionic lipid bilayers.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
-
- Varkey J.; Isas J. M.; Mizuno N.; Jensen M. B.; Bhatia V. K.; Jao C. C.; Petrlova J.; Voss J. C.; Stamou D. G.; Steven A. C.; Langen R. Membrane curvature induction and tubulation are common features of synucleins and apolipoproteins. J. Biol. Chem. 2010, 285, 32486–93. 10.1074/jbc.M110.139576. - DOI - PMC - PubMed
-
- Sharon R.; Goldberg M. S.; Bar-Josef I.; Betensky R. A.; Shen J.; Selkoe D. J. alpha-Synuclein occurs in lipid-rich high molecular weight complexes, binds fatty acids, and shows homology to the fatty acid-binding proteins. Proc. Natl. Acad. Sci. U.S.A. 2001, 98, 9110–5. 10.1073/pnas.171300598. - DOI - PMC - PubMed
-
- Adamczyk A.; Kacprzak M.; Kazmierczak A. Alpha-synuclein decreases arachidonic acid incorporation into rat striatal synaptoneurosomes. Folia Neuropathol 2007, 45 (4), 230–235. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

