Interplay of Developmental Hippo-Notch Signaling Pathways with the DNA Damage Response in Prostate Cancer
- PMID: 35954292
- PMCID: PMC9367915
- DOI: 10.3390/cells11152449
Interplay of Developmental Hippo-Notch Signaling Pathways with the DNA Damage Response in Prostate Cancer
Abstract
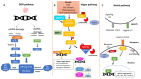
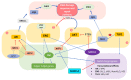
Prostate cancer belongs in the class of hormone-dependent cancers, representing a major cause of cancer incidence in men worldwide. Since upon disease onset almost all prostate cancers are androgen-dependent and require active androgen receptor (AR) signaling for their survival, the primary treatment approach has for decades relied on inhibition of the AR pathway via androgen deprivation therapy (ADT). However, following this line of treatment, cancer cell pools often become resistant to therapy, contributing to disease progression towards the significantly more aggressive castration-resistant prostate cancer (CRPC) form, characterized by poor prognosis. It is, therefore, of critical importance to elucidate the molecular mechanisms and signaling pathways underlying the progression of early-stage prostate cancer towards CRPC. In this review, we aim to shed light on the role of major signaling pathways including the DNA damage response (DDR) and the developmental Hippo and Notch pathways in prostate tumorigenesis. We recapitulate key evidence demonstrating the crosstalk of those pathways as well as with pivotal prostate cancer-related 'hubs' such as AR signaling, and evaluate the clinical impact of those interactions. Moreover, we attempt to identify molecules of the complex DDR-Hippo-Notch interplay comprising potentially novel therapeutic targets in the battle against prostate tumorigenesis.
Keywords: DNA damage response (DDR); Hippo pathway; Notch pathway; interplay; prostate cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Hååg P., Bektic J., Bartsch G., Klocker H., Eder I.E. Androgen receptor down regulation by small interference RNA induces cell growth inhibition in androgen sensitive as well as in androgen independent prostate cancer cells. J. Steroid Biochem. Mol. Biol. 2005;96:251–258. doi: 10.1016/j.jsbmb.2005.04.029. - DOI - PubMed
-
- Tan Y.G., Poon R.J., Pang L.J., Villanueva A., Huang H.H., Chen K., Ng T.K., Tay K.J., Ho H.S., Yuen J.S. Comparative study of surgical orchidectomy and medical castration in treatment efficacy, adverse effects and cost based on a large prospective metastatic prostate cancer registry. Urol. Oncol. Semin. Orig. Investig. 2020;38:682.e1–682.e9. doi: 10.1016/j.urolonc.2020.05.005. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

