Neither Tumor-Infiltrating Lymphocytes nor Cytotoxic T Cells Predict Enhanced Benefit from Chemotherapy in the DBCG77B Phase III Clinical Trial
- PMID: 35954471
- PMCID: PMC9367267
- DOI: 10.3390/cancers14153808
Neither Tumor-Infiltrating Lymphocytes nor Cytotoxic T Cells Predict Enhanced Benefit from Chemotherapy in the DBCG77B Phase III Clinical Trial
Abstract
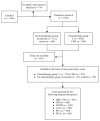
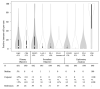
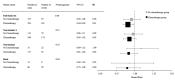
Recent studies have shown that immune infiltrates in the tumor microenvironment play a role in response to therapy, with some suggesting that patients with immunogenic tumors may receive increased benefit from chemotherapies. We evaluated this hypothesis in early breast cancer by testing the interaction between immune biomarkers and chemotherapy using materials from DBCG77B, a phase III clinical trial where high-risk premenopausal women were randomized to receive chemotherapy or no chemotherapy. Tissue microarrays were evaluated for tumor-infiltrating lymphocytes (TILs) assessed morphologically on hematoxylin and eosin-stained slides, and by immunohistochemistry for CD8, FOXP3, LAG-3, PD-1 and PD-L1. Following REMARK reporting guidelines, data analyses were performed according to a prespecified statistical plan, using 10-year invasive disease-free survival as the endpoint. Differences in survival probabilities between biomarker groups were evaluated by Kaplan-Meier and Cox proportional hazard ratio analyses and prediction for treatment benefit by an interaction test. Our results showed that stromal TILs were associated with an improved prognosis (HR = 0.93; p-value = 0.03), consistent with previous studies. However, none of the immune biomarkers predicted benefit from chemotherapy in the full study set nor within major breast cancer subtypes. Our study indicates that primary tumors with higher immune infiltration do not derive extra benefit from cyclophosphamide-based cytotoxic chemotherapy.
Keywords: adjuvant chemotherapy; breast cancer; cyclophosphamide; immuno-oncology; immunohistochemistry; lymphocyte biomarkers; tumor immune microenvironment.
Conflict of interest statement
T.O.N. has a patent for Prosigna gene expression assay, with royalties paid by licensee Veracyte and support from Bioclassifier LLC and from NanoString Technologies, all outside the submitted work. M.-B.J. has received institutional research support from NanoString Technologies, Oncology Venture, and Samsung Bioepis, all outside of the submitted work. B.E. reports institutional research support from Kræftens Bekæmpelse, Astra Zeneca, Eli Lilly, MSD, Novartis, Pfizer, Roche, and Samsung Bioepis (none of which are part of the submitted work). All remaining authors have declared no conflict of interest.
Figures

References
-
- Buccione C., Fragale A., Polverino F., Ziccheddu G., Aricò E., Belardelli F., Proietti E., Battistini A., Moschella F. Role of Interferon Regulatory Factor 1 in Governing Treg Depletion, Th1 Polarization, Inflammasome Activation and Antitumor Efficacy of Cyclophosphamide. Int. J. Cancer. 2018;142:976–987. doi: 10.1002/ijc.31083. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

