Hymenolepis diminuta Infection Affects Apoptosis in the Small and Large Intestine
- PMID: 35955110
- PMCID: PMC9368115
- DOI: 10.3390/ijerph19159753
Hymenolepis diminuta Infection Affects Apoptosis in the Small and Large Intestine
Abstract
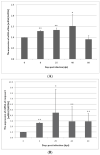
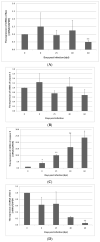
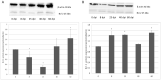
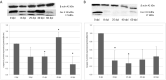
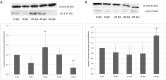
The rat tapeworm Hymenolepis diminuta has been shown to cause alterations in gastrointestinal tissues. Since hymenolepiasis induces a number of reactions in the host, it is reasonable to assume that it may also be involved in the mechanisms of apoptosis in the intestines. Individual research tasks included an examination of the effect of H. diminuta infection on; (i) the cellular localization of the expression of pro-apoptotic protein Bax and anti-apoptotic protein Bcl-2, as well as caspase-3 and caspase-9, and (ii) the effects of the infection on the expression of Bcl-2, Bax, Cas-3 and Cas-9, at the mRNA and protein levels. Molecular tests (including mRNA (qRT PCR) and the protein (Western blot) expression of Bax, Bcl-2, and caspases-3, -9) and immunohistochemical tests were performed during the experiment. They showed that H. diminuta infection activates the intrinsic apoptosis pathway in the small and large intestine of the host. H. diminuta infection triggered the apoptosis via the activation of the caspase cascade, including Cas-3 and Cas-9. Hymenolepiasis enhanced apoptosis in the small and large intestine of the host by increasing the expression of the pro-apoptotic gene and protein Bax and by decreasing the expression of the anti-apoptotic gene and protein Bcl-2.
Keywords: apoptosis; hymenolepiasis; parasite–host system; rat.
Conflict of interest statement
The authors declare no conflict of interest.
Figures










Similar articles
-
Hymenolepis diminuta: analysis of the expression of Toll-like receptor genes and protein (TLR3 and TLR9) in the small and large intestines of rats.Exp Parasitol. 2014 Oct;145:61-7. doi: 10.1016/j.exppara.2014.07.009. Epub 2014 Aug 1. Exp Parasitol. 2014. PMID: 25092440
-
Hymenolepis diminuta: analysis of the expression of Toll-like receptor genes (TLR2 and TLR4) in the small and large intestines of rats. Part II.Exp Parasitol. 2013 Oct;135(2):437-45. doi: 10.1016/j.exppara.2013.08.002. Epub 2013 Aug 27. Exp Parasitol. 2013. PMID: 23994484
-
Hymenolepis diminuta fractions but not previous tapeworm infection stimulate intestinal myoelectric alterations in vivo in the rat.J Parasitol. 1998 Aug;84(4):673-80. J Parasitol. 1998. PMID: 9714192
-
Selected Molecular Mechanisms Involved in the Parasite⁻Host System Hymenolepis diminuta⁻Rattus norvegicus.Int J Mol Sci. 2018 Aug 17;19(8):2435. doi: 10.3390/ijms19082435. Int J Mol Sci. 2018. PMID: 30126154 Free PMC article. Review.
-
Worldwide overview of human infections with Hymenolepis diminuta.Parasitol Res. 2020 Jul;119(7):1997-2004. doi: 10.1007/s00436-020-06663-x. Epub 2020 Mar 24. Parasitol Res. 2020. PMID: 32211990 Review.
Cited by
-
The Tapeworm Hymenolepis diminuta as an Important Model Organism in the Experimental Parasitology of the 21st Century.Pathogens. 2022 Nov 29;11(12):1439. doi: 10.3390/pathogens11121439. Pathogens. 2022. PMID: 36558772 Free PMC article. Review.
References
-
- Kapczuk P., Chlubek D., Baranowska-Bosiacka I. Epidemiological and clinical characteristic of Hymenolepis diminuta infection—review of current literature. Pomeranian J. Life Sci. 2020;66:32–38. doi: 10.21164/pomjlifesci.664. - DOI
-
- Fal W., Czaplicka H. Effect of experimental hymenolepiasis on various tissue reactions in rats. Wiad. Parazytol. 1991;37:331–342. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

