Chlorhexidine Promotes Psl Expression in Pseudomonas aeruginosa That Enhances Cell Aggregation with Preserved Pathogenicity Demonstrates an Adaptation against Antiseptic
- PMID: 35955437
- PMCID: PMC9368580
- DOI: 10.3390/ijms23158308
Chlorhexidine Promotes Psl Expression in Pseudomonas aeruginosa That Enhances Cell Aggregation with Preserved Pathogenicity Demonstrates an Adaptation against Antiseptic
Abstract
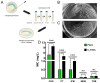
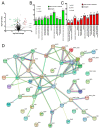
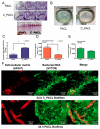
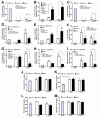
Because Pseudomonas aeruginosa is frequently in contact with Chlorhexidine (a regular antiseptic), bacterial adaptations are possible. In comparison with the parent strain, the Chlorhexidine-adapted strain formed smaller colonies with metabolic downregulation (proteomic analysis) with the cross-resistance against colistin (an antibiotic for several antibiotic-resistant bacteria), partly through the modification of L-Ara4N in the lipopolysaccharide at the outer membrane. Chlorhexidine-adapted strain formed dense liquid-solid interface biofilms with enhanced cell aggregation partly due to the Chlorhexidine-induced overexpression of psl (exopolysaccharide-encoded gene) through the LadS/GacSA pathway (c-di-GMP-independence) in 12 h biofilms and maintained the aggregation with SiaD-mediated c-di-GMP dependence in 24 h biofilms as evaluated by polymerase chain reaction (PCR). The addition of Ca2+ in the Chlorhexidine-adapted strain facilitated several Psl-associated genes, indicating an impact of Ca2+ in Psl production. The activation by Chlorhexidine-treated sessile bacteria demonstrated a lower expression of IL-6 and IL-8 on fibroblasts and macrophages than the activation by the parent strain, indicating the less inflammatory reactions from Chlorhexidine-exposed bacteria. However, the 14-day severity of the wounds in mouse caused by Chlorhexidine-treated bacteria versus the parent strain was similar, as indicated by wound diameters and bacterial burdens. In conclusion, Chlorhexidine induced psl over-expression and colistin cross-resistance that might be clinically important.
Keywords: Pseudomonas aeruginosa; Psl; biofilms; cell aggregate; chlorhexidine; cross-resistance; wound.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures




Similar articles
-
Matrix Polysaccharides and SiaD Diguanylate Cyclase Alter Community Structure and Competitiveness of Pseudomonas aeruginosa during Dual-Species Biofilm Development with Staphylococcus aureus.mBio. 2018 Nov 6;9(6):e00585-18. doi: 10.1128/mBio.00585-18. mBio. 2018. PMID: 30401769 Free PMC article.
-
Pseudomonas aeruginosa uses a cyclic-di-GMP-regulated adhesin to reinforce the biofilm extracellular matrix.Mol Microbiol. 2010 Feb;75(4):827-42. doi: 10.1111/j.1365-2958.2009.06991.x. Epub 2010 Jan 17. Mol Microbiol. 2010. PMID: 20088866 Free PMC article.
-
Mucoid Pseudomonas aeruginosa Can Produce Calcium-Gelled Biofilms Independent of the Matrix Components Psl and CdrA.J Bacteriol. 2022 May 17;204(5):e0056821. doi: 10.1128/jb.00568-21. Epub 2022 Apr 13. J Bacteriol. 2022. PMID: 35416688 Free PMC article.
-
Recent perspectives on the molecular basis of biofilm formation by Pseudomonas aeruginosa and approaches for treatment and biofilm dispersal.Folia Microbiol (Praha). 2018 Jul;63(4):413-432. doi: 10.1007/s12223-018-0585-4. Epub 2018 Jan 19. Folia Microbiol (Praha). 2018. PMID: 29352409 Review.
-
Susceptibility of antibiotic-resistant Gram-negative bacteria to biocides: a perspective from the study of catheter biofilms.J Appl Microbiol. 2002;92 Suppl:163S-70S. J Appl Microbiol. 2002. PMID: 12000625 Review.
Cited by
-
Lacticaseibacillus rhamnosus dfa1 Attenuate Cecal Ligation-Induced Systemic Inflammation through the Interference in Gut Dysbiosis, Leaky Gut, and Enterocytic Cell Energy.Int J Mol Sci. 2023 Feb 13;24(4):3756. doi: 10.3390/ijms24043756. Int J Mol Sci. 2023. PMID: 36835163 Free PMC article.
-
Biofilms as protective cocoons against biocides: from bacterial adaptation to One Health issues.Microbiology (Reading). 2023 Jun;169(6):001340. doi: 10.1099/mic.0.001340. Microbiology (Reading). 2023. PMID: 37266984 Free PMC article. Review.
-
Hydrogen peroxide from L-amino acid oxidase of king cobra (Ophiophagus hannah) venom attenuates Pseudomonas biofilms.Sci Rep. 2023 Jul 12;13(1):11304. doi: 10.1038/s41598-023-37914-3. Sci Rep. 2023. PMID: 37438396 Free PMC article.
-
The Regulatory Roles of Ezh2 in Response to Lipopolysaccharide (LPS) in Macrophages and Mice with Conditional Ezh2 Deletion with LysM-Cre System.Int J Mol Sci. 2023 Mar 10;24(6):5363. doi: 10.3390/ijms24065363. Int J Mol Sci. 2023. PMID: 36982437 Free PMC article.
-
Bacteriophages isolated from mouse feces attenuates pneumonia mice caused by Pseudomonas aeruginosa.PLoS One. 2024 Jul 16;19(7):e0307079. doi: 10.1371/journal.pone.0307079. eCollection 2024. PLoS One. 2024. PMID: 39012882 Free PMC article.
References
MeSH terms
Substances
Grants and funding
- CUFRB65_hea [33] _040_30_21/Chulalongkorn University through Fundamental Fund 65
- B16F640175 and B05F640144/the National Research Council of Thailand (811/2563) with NSRF via the Program Management Unit for Human Resources & Institutional Development, Research and Innovation
- the Second Century Fund (C2F) for Postdoctoral Fellowship, Chulalongkorn University
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

