Electron Attachment to 5-Fluorouracil: The Role of Hydrogen Fluoride in Dissociation Chemistry
- PMID: 35955461
- PMCID: PMC9369043
- DOI: 10.3390/ijms23158325
Electron Attachment to 5-Fluorouracil: The Role of Hydrogen Fluoride in Dissociation Chemistry
Abstract
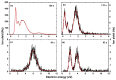
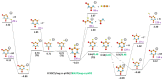
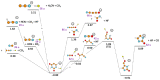
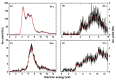
We investigate dissociative electron attachment to 5-fluorouracil (5-FU) employing a crossed electron-molecular beam experiment and quantum chemical calculations. Upon the formation of the 5-FU- anion, 12 different fragmentation products are observed, the most probable dissociation channel being H loss. The parent anion, 5-FU-, is not stable on the experimental timescale (~140 µs), most probably due to the low electron affinity of FU; simple HF loss and F- formation are seen only with a rather weak abundance. The initial dynamics upon electron attachment seems to be governed by hydrogen atom pre-dissociation followed by either its full dissociation or roaming in the vicinity of the molecule, recombining eventually into the HF molecule. When the HF molecule is formed, the released energy might be used for various ring cleavage reactions. Our results show that higher yields of the fluorine anion are most probably prevented through both faster dissociation of an H atom and recombination of F- with a proton to form HF. Resonance calculations indicate that F- is formed upon shape as well as core-excited resonances.
Keywords: 5-fluorouracil; anion; electron attachment; hydrogen fluoride.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Gorfinkiel J.D., Ptasinska S. Electron Scattering from Molecules and Molecular Aggregates of Biological Relevance. J. Phys. B At. Mol. Opt. Phys. 2017;50:182001. doi: 10.1088/1361-6455/aa8572. - DOI
-
- Abouaf R., Dunet H. Structures in Dissociative Electron Attachment Cross-Sections in Thymine, Uracil and Halouracils. Eur. Phys. J. D. 2005;35:405–410. doi: 10.1140/epjd/e2005-00239-9. - DOI
-
- Abdoul-Carime H., Huels M.A., Illenberger E., Sanche L. Formation of Negative Ions from Gas Phase Halo-Uracils by Low-Energy (0–18 eV) Electron Impact. Int. J. Mass Spectrom. 2003;228:703–716. doi: 10.1016/S1387-3806(03)00139-8. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

