Inhibition of Monoacylglycerol Lipase by NSD1819 as an Effective Strategy for the Endocannabinoid System Modulation against Neuroinflammation-Related Disorders
- PMID: 35955562
- PMCID: PMC9369272
- DOI: 10.3390/ijms23158428
Inhibition of Monoacylglycerol Lipase by NSD1819 as an Effective Strategy for the Endocannabinoid System Modulation against Neuroinflammation-Related Disorders
Abstract
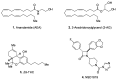
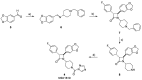
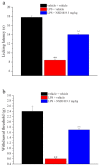
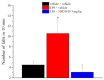
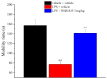
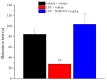
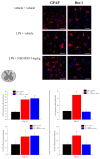
Neuroinflammation is a key pathological event shared by different diseases affecting the nervous system. Since the underlying mechanism of neuroinflammation is a complex and multifaceted process, current pharmacological treatments are unsatisfactory-a reason why new therapeutic approaches are mandatory. In this context, the endocannabinoid system has proven to possess neuroprotective and immunomodulatory actions under neuroinflammatory status, and its modulation could represent a valuable approach to address different inflammatory processes. To this aim, we evaluated the efficacy of a repeated treatment with NSD1819, a potent β-lactam-based monoacylglycerol lipase inhibitor in a mouse model of neuroinflammation induced by lipopolysaccharide (LPS) injection. Mice were intraperitoneally injected with LPS 1 mg/kg for five consecutive days to induce systemic inflammation. Concurrently, NSD1819 (3 mg/kg) was daily per os administered from day 1 until the end of the experiment (day 11). Starting from day 8, behavioral measurements were performed to evaluate the effect of the treatment on cognitive impairments, allodynia, motor alterations, anhedonia, and depressive-like behaviors evoked by LPS. Histologically, glial analysis of the spinal cord was also performed. The administration of NSD1819 was able to completely counteract thermal and mechanical allodynia as highlighted by the Cold plate and von Frey tests, respectively, and to reduce motor impairments as demonstrated by the Rota rod test. Moreover, the compound was capable of neutralizing the memory loss in the Passive avoidance test, and reducing depressive-like behavior in the Porsolt test. Finally, LPS stimulation caused a significant glial cells activation in the dorsal horn of the lumbar spinal cord that was significantly recovered by NSD1819 repeated treatment. In conclusion, NSD1819 was able to thwart the plethora of symptoms evoked by LPS, thus representing a promising candidate for future applications in the context of neuroinflammation and related diseases.
Keywords: 2-AG; LPS; MGL inhibitor; depression; glial cells; memory; neuroinflammation; pain.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Efficacy of a vegetal mixture composed of Zingiber officinale, Echinacea purpurea, and Centella asiatica in a mouse model of neuroinflammation: In vivo and ex vivo analysis.Front Nutr. 2022 Aug 30;9:887378. doi: 10.3389/fnut.2022.887378. eCollection 2022. Front Nutr. 2022. PMID: 36118773 Free PMC article.
-
Alterations in endocannabinoid tone following chemotherapy-induced peripheral neuropathy: effects of endocannabinoid deactivation inhibitors targeting fatty-acid amide hydrolase and monoacylglycerol lipase in comparison to reference analgesics following cisplatin treatment.Pharmacol Res. 2013 Jan;67(1):94-109. doi: 10.1016/j.phrs.2012.10.013. Epub 2012 Nov 2. Pharmacol Res. 2013. PMID: 23127915 Free PMC article.
-
Endocannabinoid modulation of inflammatory hyperalgesia in the IFN-α mouse model of depression.Brain Behav Immun. 2019 Nov;82:372-381. doi: 10.1016/j.bbi.2019.09.006. Epub 2019 Sep 7. Brain Behav Immun. 2019. PMID: 31505257
-
Sleep, Glial Function, and the Endocannabinoid System: Implications for Neuroinflammation and Sleep Disorders.Int J Mol Sci. 2024 Mar 9;25(6):3160. doi: 10.3390/ijms25063160. Int J Mol Sci. 2024. PMID: 38542134 Free PMC article. Review.
-
Environmental Enrichment Protects against Neurotoxic Effects of Lipopolysaccharide: A Comprehensive Overview.Int J Mol Sci. 2023 Mar 11;24(6):5404. doi: 10.3390/ijms24065404. Int J Mol Sci. 2023. PMID: 36982478 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

