Highly Organized Porous Gelatin-Based Scaffold by Microfluidic 3D-Foaming Technology and Dynamic Culture for Cartilage Tissue Engineering
- PMID: 35955581
- PMCID: PMC9369316
- DOI: 10.3390/ijms23158449
Highly Organized Porous Gelatin-Based Scaffold by Microfluidic 3D-Foaming Technology and Dynamic Culture for Cartilage Tissue Engineering
Abstract
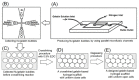
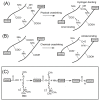
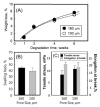
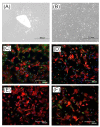
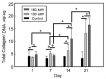
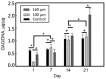
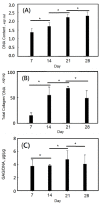
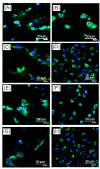
A gelatin-based hydrogel scaffold with highly uniform pore size and biocompatibility was fabricated for cartilage tissue engineering using microfluidic 3D-foaming technology. Mainly, bubbles with different diameters, such as 100 μm and 160 μm, were produced by introducing an optimized nitrogen gas and gelatin solution at an optimized flow rate, and N2/gelatin bubbles were formed. Furthermore, a cross-linking agent (1-ethyl-3-(3-dimethyl aminopropyl)-carbodiimide, EDC) was employed for the cross-linking reaction of the gelatin-based hydrogel scaffold with uniform bubbles, and then the interface between the close cells were broken by degassing. The pore uniformity of the gelatin-based hydrogel scaffolds was confirmed by use of a bright field microscope, conjugate focus microscope and scanning electron microscope. The in vitro degradation rate, mechanical properties, and swelling rate of gelatin-based hydrogel scaffolds with highly uniform pore size were studied. Rabbit knee cartilage was cultured, and its extracellular matrix content was analyzed. Histological analysis and immunofluorescence staining were employed to confirm the activity of the rabbit knee chondrocytes. The chondrocytes were seeded into the resulting 3D porous gelatin-based hydrogel scaffolds. The growth conditions of the chondrocyte culture on the resulting 3D porous gelatin-based hydrogel scaffolds were evaluated by MTT analysis, live/dead cell activity analysis, and extracellular matrix content analysis. Additionally, a dynamic culture of cartilage tissue was performed, and the expression of cartilage-specific proteins within the culture time was studied by immunofluorescence staining analysis. The gelatin-based hydrogel scaffold encouraged chondrocyte proliferation, promoting the expression of collagen type II, aggrecan, and sox9 while retaining the structural stability and durability of the cartilage after dynamic compression and promoting cartilage repair.
Keywords: cartilage tissue engineering; dynamic culture; gelatin; microfluidics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






Similar articles
-
3D-Printed PEG-PLA/Gelatin Hydrogel: Characterization toward In Vitro Chondrocyte Redifferentiation.ACS Biomater Sci Eng. 2025 Apr 14;11(4):2157-2166. doi: 10.1021/acsbiomaterials.4c02409. Epub 2025 Mar 19. ACS Biomater Sci Eng. 2025. PMID: 40108844 Free PMC article.
-
[Research of diclofenac sodium-loaded gelatin scaffold with anti-inflammatory activity for promoting in vivo cartilage regeneration].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2023 Jan 15;37(1):91-100. doi: 10.7507/1002-1892.202207114. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2023. PMID: 36708121 Free PMC article. Chinese.
-
Synthesis and in vitro evaluation of thermosensitive hydrogel scaffolds based on (PNIPAAm-PCL-PEG-PCL-PNIPAAm)/Gelatin and (PCL-PEG-PCL)/Gelatin for use in cartilage tissue engineering.J Biomater Sci Polym Ed. 2018 Jul;29(10):1185-1206. doi: 10.1080/09205063.2018.1447627. Epub 2018 Mar 5. J Biomater Sci Polym Ed. 2018. PMID: 29490569
-
Anatomy, molecular structures, and hyaluronic acid - Gelatin injectable hydrogels as a therapeutic alternative for hyaline cartilage recovery: A review.J Biomed Mater Res B Appl Biomater. 2023 Sep;111(9):1705-1722. doi: 10.1002/jbm.b.35261. Epub 2023 May 13. J Biomed Mater Res B Appl Biomater. 2023. PMID: 37178328 Review.
-
Progress of Microfluidic Hydrogel-Based Scaffolds and Organ-on-Chips for the Cartilage Tissue Engineering.Adv Mater. 2023 Jun;35(26):e2208852. doi: 10.1002/adma.202208852. Epub 2023 May 8. Adv Mater. 2023. PMID: 36633376 Review.
Cited by
-
Gradient scaffolds in bone-soft tissue interface engineering: Structural characteristics, fabrication techniques, and emerging trends.J Orthop Translat. 2025 Jan 28;50:333-353. doi: 10.1016/j.jot.2024.10.015. eCollection 2025 Jan. J Orthop Translat. 2025. PMID: 39944791 Free PMC article. Review.
-
Hemocompatible gelatin-glycidyl methacrylate/graphene oxide composite hydrogels for vascular catheter applications.Sci Rep. 2025 Mar 25;15(1):10224. doi: 10.1038/s41598-025-93040-2. Sci Rep. 2025. PMID: 40133392 Free PMC article.
-
Controlling Microenvironments with Organs-on-Chips for Osteoarthritis Modelling.Cells. 2023 Feb 10;12(4):579. doi: 10.3390/cells12040579. Cells. 2023. PMID: 36831245 Free PMC article. Review.
-
Digital light processing printing of non-modified protein-only compositions.Mater Today Bio. 2024 Dec 6;30:101384. doi: 10.1016/j.mtbio.2024.101384. eCollection 2025 Feb. Mater Today Bio. 2024. PMID: 39790486 Free PMC article.
-
Preparation of hydrogel microsphere and its application in articular cartilage injury.Mater Today Bio. 2025 Mar 8;31:101641. doi: 10.1016/j.mtbio.2025.101641. eCollection 2025 Apr. Mater Today Bio. 2025. PMID: 40130039 Free PMC article. Review.
References
-
- Liaw D.J., Huang C.C., Sang H.C., Kang E.T. Intramolecular hydrophobic aggregation of amphiphilic polysulfobetaine with Various Hydrophobic Groups in Aqueous Solution. Langmuir. 1999;15:5204–5211. doi: 10.1021/la980728h. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

