Psychological Stress Exacerbates Inflammation of the Ileum via the Corticotropin-Releasing Hormone-Mast Cell Axis in a Mouse Model of Eosinophilic Enteritis
- PMID: 35955675
- PMCID: PMC9369025
- DOI: 10.3390/ijms23158538
Psychological Stress Exacerbates Inflammation of the Ileum via the Corticotropin-Releasing Hormone-Mast Cell Axis in a Mouse Model of Eosinophilic Enteritis
Abstract
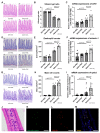
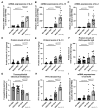
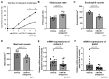
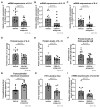
The effects of psychological stress on eosinophilic gastrointestinal disorders have not been elucidated. This study investigated the effects of psychological stress in a mouse model of eosinophilic enteritis (EoN). BALB/c mice were treated with ovalbumin (OVA) to create an EoN model and subjected to either water avoidance stress (WAS) or sham stress (SS). Microscopic inflammation, eosinophil and mast cell counts, mRNA expression, and protein levels of type 2 helper T cell (Th2) cytokines in the ileum were compared between groups. We evaluated ex vivo intestinal permeability using an Ussing chamber. A corticotropin-releasing hormone type 1 receptor (CRH-R1) antagonist was administered before WAS, and its effects were analyzed. WAS significantly increased diarrhea occurrence and, eosinophil and mast cell counts, and decreased the villus/crypt ratio compared to those in the SS group. The mRNA expression of CRH, interleukin IL-4, IL-5, IL-13, eotaxin-1, and mast cell tryptase β2 significantly increased, and the protein levels of IL-5, IL-13, and OVA-specific immunoglobulin E (IgE) also significantly increased in the WAS group. Moreover, WAS significantly increased the intestinal permeability. The CRH-R1 antagonist significantly inhibited all changes induced by WAS. Psychological stress exacerbated ileal inflammation via the CRH-mast cell axis in an EoN mouse model.
Keywords: corticotropin-releasing hormone; eosinophilic enteritis; eosinophilic gastrointestinal disorders; eosinophils; intestinal permeability; mast cells; psychological stress.
Conflict of interest statement
Y.F. had grant support from DAIICHI SANKYO COMPANY, LIMITED (Tokyo, Japan) and EA Pharma Co., Ltd (Tokyo, Japan).
Figures

Similar articles
-
Regulation of corticotropin-releasing hormone receptor-2 expression in human cord blood-derived cultured mast cells.J Mol Endocrinol. 2005 Dec;35(3):R1-8. doi: 10.1677/jme.1.01833. J Mol Endocrinol. 2005. PMID: 16326828
-
Fish oil enhances intestinal barrier function and inhibits corticotropin-releasing hormone/corticotropin-releasing hormone receptor 1 signalling pathway in weaned pigs after lipopolysaccharide challenge.Br J Nutr. 2016 Jun;115(11):1947-57. doi: 10.1017/S0007114516001100. Epub 2016 Apr 15. Br J Nutr. 2016. PMID: 27080003
-
Corticotropin-releasing hormone (CRH) regulates macromolecular permeability via mast cells in normal human colonic biopsies in vitro.Gut. 2008 Jan;57(1):50-8. doi: 10.1136/gut.2006.117549. Epub 2007 May 24. Gut. 2008. PMID: 17525093
-
Corticotropin-releasing hormone-binding protein and stress: from invertebrates to humans.Stress. 2017 Sep;20(5):449-464. doi: 10.1080/10253890.2017.1322575. Epub 2017 May 18. Stress. 2017. PMID: 28436309 Free PMC article. Review.
-
Contribution of stress to asthma worsening through mast cell activation.Ann Allergy Asthma Immunol. 2012 Jul;109(1):14-9. doi: 10.1016/j.anai.2012.03.003. Epub 2012 Mar 22. Ann Allergy Asthma Immunol. 2012. PMID: 22727152 Review.
Cited by
-
Chronic variable stress leads to sex specific gut microbiome alterations in mice.Brain Behav Immun Health. 2024 Mar 21;37:100755. doi: 10.1016/j.bbih.2024.100755. eCollection 2024 May. Brain Behav Immun Health. 2024. PMID: 38618010 Free PMC article.
-
Depression Pathophysiology: Astrocyte Mitochondrial Melatonergic Pathway as Crucial Hub.Int J Mol Sci. 2022 Dec 26;24(1):350. doi: 10.3390/ijms24010350. Int J Mol Sci. 2022. PMID: 36613794 Free PMC article. Review.
-
Correlation between anxiety and decreased quality of life in patients with non-esophageal eosinophilic gastrointestinal diseases.JGH Open. 2023 Dec 23;8(1):e13025. doi: 10.1002/jgh3.13025. eCollection 2024 Jan. JGH Open. 2023. PMID: 38268958 Free PMC article.
-
The Association between Functional Dyspepsia and Metabolic Syndrome-The State of the Art.Int J Environ Res Public Health. 2024 Feb 18;21(2):237. doi: 10.3390/ijerph21020237. Int J Environ Res Public Health. 2024. PMID: 38397726 Free PMC article. Review.
References
-
- Dellon E.S., Gonsalves N., Abonia J.P., Alexander J.A., Arva N.C., Atkins D., Attwood S.E., Auth M.K.H., Bailey D.D., Biederman L., et al. International Consensus Recommendations for Eosinophilic Gastrointestinal Disease Nomenclature. Clin. Gastroenterol. Hepatol. 2022 doi: 10.1016/j.cgh.2022.02.017. - DOI - PMC - PubMed
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources

