Application of Perinatal Derivatives on Oncological Preclinical Models: A Review of Animal Studies
- PMID: 35955703
- PMCID: PMC9369310
- DOI: 10.3390/ijms23158570
Application of Perinatal Derivatives on Oncological Preclinical Models: A Review of Animal Studies
Abstract
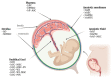
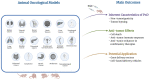
The increasing cancer incidence has certified oncological management as one of the most critical challenges for the coming decades. New anticancer strategies are still needed, despite the significant advances brought to the forefront in the last decades. The most recent, promising therapeutic approaches have benefitted from the application of human perinatal derivatives (PnD), biological mediators with proven benefits in several fields beyond oncology. To elucidate preclinical results and clinic outcomes achieved in the oncological field, we present a narrative review of the studies resorting to animal models to assess specific outcomes of PnD products. Recent preclinical evidence points to promising anticancer effects offered by PnD mediators isolated from the placenta, amniotic membrane, amniotic fluid, and umbilical cord. Described effects include tumorigenesis prevention, uncontrolled growth or regrowth inhibition, tumor homing ability, and adequate cell-based delivery capacity. Furthermore, PnD treatments have been described as supportive of chemotherapy and radiological therapies, particularly when resistance has been reported. However, opposite effects of PnD products have also been observed, offering support and trophic effect to malignant cells. Such paradoxical and dichotomous roles need to be intensively investigated. Current hypotheses identify as explanatory some critical factors, such as the type of the PnD biological products used or the manufacturing procedure to prepare the tissue/cellular treatment, the experimental design (including human-relevant animal models), and intrinsic pathophysiological characteristics. The effective and safe translation of PnD treatments to clinical practice relies on the collaborative efforts of all researchers working with human-relevant oncological preclinical models. However, it requires proper guidelines and consensus compiled by experts and health workers who accurately describe the methodology of tissue collection, PnD isolation, manufacturing, preservation, and delivery to the final user.
Keywords: animal models; cancer; perinatal derivatives; preclinical studies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- World Health Organization . WHO Report on Cancer: Setting Priorities, Investing Wisely and Providing Care for All. WHO; Geneva, Switzerland: 2020.
-
- Bidram E., Esmaeili Y., Ranji-Burachaloo H., Al-Zaubai N., Zarrabi A., Stewart A., Dunstan D.E. A concise review on cancer treatment methods and delivery systems. J. Drug Deliv. Sci. Technol. 2019;54:101350. doi: 10.1016/j.jddst.2019.101350. - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

