Targeting of a Conserved Epitope in Mouse and Human GPVI Differently Affects Receptor Function
- PMID: 35955743
- PMCID: PMC9369317
- DOI: 10.3390/ijms23158610
Targeting of a Conserved Epitope in Mouse and Human GPVI Differently Affects Receptor Function
Abstract
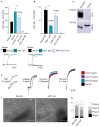
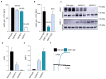
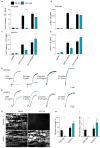
Glycoprotein (GP) VI is the major platelet collagen receptor and a promising anti-thrombotic target. This was first demonstrated in mice using the rat monoclonal antibody JAQ1, which completely blocks the Collagen-Related Peptide (CRP)-binding site on mouse GPVI and efficiently inhibits mouse platelet adhesion, activation and aggregation on collagen. Here, we show for the first time that JAQ1 cross-reacts with human GPVI (huGPVI), but not with GPVI in other tested species, including rat, rabbit, guinea pig, swine, and dog. We further demonstrate that JAQ1 differently modulates mouse and human GPVI function. Similar to its effects on mouse GPVI (mGPVI), JAQ1 inhibits CRP-induced activation in human platelets, whereas, in stark contrast to mouse GPVI, it does not inhibit the adhesion, activation or aggregate formation of human platelets on collagen, but causes instead an increased response. This effect was also seen with platelets from newly generated human GPVI knockin mice (hGP6tg/tg). These results indicate that the binding of JAQ1 to a structurally conserved epitope in GPVI differently affects its function in human and mouse platelets.
Keywords: JAQ1; glycoprotein VI; platelet activation; platelet inhibition; platelet receptors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Evidence for two distinct epitopes within collagen for activation of murine platelets.J Biol Chem. 2001 Jan 5;276(1):364-8. doi: 10.1074/jbc.M007536200. J Biol Chem. 2001. PMID: 11036078
-
Targeting of the collagen-binding site on glycoprotein VI is not essential for in vivo depletion of the receptor.Blood. 2003 May 15;101(10):3948-52. doi: 10.1182/blood-2002-10-3242. Epub 2003 Jan 16. Blood. 2003. PMID: 12531795
-
Expression and function of the mouse collagen receptor glycoprotein VI is strictly dependent on its association with the FcRgamma chain.J Biol Chem. 2000 Aug 4;275(31):23998-4002. doi: 10.1074/jbc.M003803200. J Biol Chem. 2000. PMID: 10825177
-
Clinical significance of altered collagen-receptor functioning in platelets with emphasis on glycoprotein VI.Blood Rev. 2019 Nov;38:100592. doi: 10.1016/j.blre.2019.100592. Epub 2019 Jul 22. Blood Rev. 2019. PMID: 31351674 Review.
-
[Advances in the studies of platelet glycoprotein VI (GPVI): review].Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2006 Oct;14(5):1040-4. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2006. PMID: 17096916 Review. Chinese.
Cited by
-
Contrasting Effects of Platelet GPVI Deletion Versus Syk Inhibition on Mouse Jugular Vein Puncture Wound Structure.Int J Mol Sci. 2025 May 1;26(9):4294. doi: 10.3390/ijms26094294. Int J Mol Sci. 2025. PMID: 40362537 Free PMC article.
-
Minimal Collagen-Binding Epitope of Glycoprotein VI in Human and Mouse Platelets.Biomedicines. 2023 Feb 1;11(2):423. doi: 10.3390/biomedicines11020423. Biomedicines. 2023. PMID: 36830959 Free PMC article.
-
The humanized platelet glycoprotein VI Fab inhibitor EMA601 protects from arterial thrombosis and ischaemic stroke in mice.Eur Heart J. 2024 Nov 14;45(43):4582-4597. doi: 10.1093/eurheartj/ehae482. Eur Heart J. 2024. PMID: 39150906 Free PMC article.
-
Divalent nanobodies to platelet CLEC-2 can serve as agonists or antagonists.Commun Biol. 2023 Apr 7;6(1):376. doi: 10.1038/s42003-023-04766-6. Commun Biol. 2023. PMID: 37029319 Free PMC article.
References
-
- Michelson A.D., Cattaneo M., Frelinger A., Newman P. Platelets. 4th ed. Elsevier; Amsterdam, The Netherlands: 2019.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

