Phytoecdysteroids: Distribution, Structural Diversity, Biosynthesis, Activity, and Crosstalk with Phytohormones
- PMID: 35955797
- PMCID: PMC9369314
- DOI: 10.3390/ijms23158664
Phytoecdysteroids: Distribution, Structural Diversity, Biosynthesis, Activity, and Crosstalk with Phytohormones
Abstract
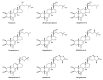
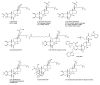
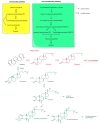
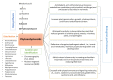
Phytoecdysteroids (PEs) are naturally occurring polyhydroxylated compounds with a structure similar to that of insect molting hormone and the plant hormone brassinosteroids. PEs have a four-ringed skeleton composed of 27, 28, 29, or 30 carbon atoms (derived from plant sterols). The carbon skeleton of ecdysteroid is known as cyclopentanoperhydrophenanthrene and has a β-sidechain on C-17. Plants produce PEs via the mevalonate pathway with the help of the precursor acetyl-CoA. PEs are found in algae, fungi, ferns, gymnosperms, and angiosperms; more than 500 different PEs are found in over 100 terrestrial plants. 20-hydroxyecdysone is the most common PE. PEs exhibit versatile biological roles in plants, invertebrates, and mammals. These compounds contribute to mitigating biotic and abiotic stresses. In plants, PEs play a potent role in enhancing tolerance against insects and nematodes via their allelochemical activity, which increases plant biological and metabolic responses. PEs promote enzymatic and non-enzymatic antioxidant defense systems, which decrease reactive oxygen species in the form of superoxide radicals and hydroxyl radicals and reduce malondialdehyde content. PEs also induce protein biosynthesis and modulate carbohydrate and lipid synthesis. In humans, PEs display biological, pharmacological, and medicinal properties, such as anti-diabetic, antioxidant, anti-microbial, hepatoprotective, hypoglycemic, anti-cancer, anti-inflammatory, antidepressant, and tissue differentiation activity.
Keywords: allelopathy; anti-stress activity; germination; growth regulators; heavy metals; isolation; pathogen; salinity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Plant ecdysteroids: plant sterols with intriguing distributions, biological effects and relations to plant hormones.Planta. 2016 Sep;244(3):545-55. doi: 10.1007/s00425-016-2561-z. Epub 2016 Jun 23. Planta. 2016. PMID: 27339274 Review.
-
The phytochemical, biological, and medicinal attributes of phytoecdysteroids: An updated review.Acta Pharm Sin B. 2021 Jul;11(7):1740-1766. doi: 10.1016/j.apsb.2020.10.012. Epub 2020 Oct 16. Acta Pharm Sin B. 2021. PMID: 34386319 Free PMC article. Review.
-
Plant hormone-mediated regulation of stress responses.BMC Plant Biol. 2016 Apr 14;16:86. doi: 10.1186/s12870-016-0771-y. BMC Plant Biol. 2016. PMID: 27079791 Free PMC article. Review.
-
Biosynthesis and distribution of insect-molting hormones in plants--a review.Lipids. 1995 Mar;30(3):257-62. doi: 10.1007/BF02537830. Lipids. 1995. PMID: 7791535 Review.
-
Function and Mechanism of Jasmonic Acid in Plant Responses to Abiotic and Biotic Stresses.Int J Mol Sci. 2021 Aug 9;22(16):8568. doi: 10.3390/ijms22168568. Int J Mol Sci. 2021. PMID: 34445272 Free PMC article. Review.
Cited by
-
Ecdysteroids from the Korean Endemic Species Ajuga spectabilis with Activities against Glucocorticoid Receptors and 11β-Hydroxysteroid Dehydrogenase Type 1.ACS Omega. 2023 Jul 13;8(29):26191-26200. doi: 10.1021/acsomega.3c02421. eCollection 2023 Jul 25. ACS Omega. 2023. PMID: 37521666 Free PMC article.
-
Ecdysterone and Turkesterone-Compounds with Prominent Potential in Sport and Healthy Nutrition.Nutrients. 2024 May 2;16(9):1382. doi: 10.3390/nu16091382. Nutrients. 2024. PMID: 38732627 Free PMC article. Review.
-
Molecular Targets of 20-Hydroxyecdysone in Mammals, Mechanism of Action: Is It a Calorie Restriction Mimetic and Anti-Aging Compound?Cells. 2025 Mar 13;14(6):431. doi: 10.3390/cells14060431. Cells. 2025. PMID: 40136680 Free PMC article. Review.
-
Phytoecdysteroids: Quantification in Selected Plant Species and Evaluation of Some Effects on Gastric Smooth Muscles.Molecules. 2024 Oct 31;29(21):5145. doi: 10.3390/molecules29215145. Molecules. 2024. PMID: 39519789 Free PMC article.
-
β-ecdysone/PLGA composite scaffolds promote skull defect healing in diabetic rat.Front Bioeng Biotechnol. 2025 Jan 13;12:1536102. doi: 10.3389/fbioe.2024.1536102. eCollection 2024. Front Bioeng Biotechnol. 2025. PMID: 39872465 Free PMC article.
References
-
- Butenandt A., Karlson P. Über die isolierung eines metamorphose-hormons der insekten in kristallisierter form. Z. Naturforsch. B. 1954;9:389–391. doi: 10.1515/znb-1954-0601. - DOI
-
- Dinan L., Harmatha J., Volodin V., Lafont R. Phytoecdysteroids: Diversity, biosynthesis and distribution. In: Smagghe G., editor. Ecdysone: Structures and Functions. Springer; Dordrecht, The Netherland: 2009. pp. 3–45. - DOI
-
- Hikino H., Takemoto T. Ecdysones of plant origin. In: Burdette W.J., editor. Invertebrate Endocrinology and Hormonal Heterophylly. Springer; Berlin/Heidelberg, Germany: 1974. pp. 185–203. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

