Roles of Focal Adhesion Kinase PTK2 and Integrin αIIbβ3 Signaling in Collagen- and GPVI-Dependent Thrombus Formation under Shear
- PMID: 35955827
- PMCID: PMC9369275
- DOI: 10.3390/ijms23158688
Roles of Focal Adhesion Kinase PTK2 and Integrin αIIbβ3 Signaling in Collagen- and GPVI-Dependent Thrombus Formation under Shear
Abstract
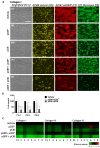
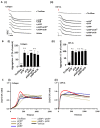
Glycoprotein (GP)VI and integrin αIIbβ3 are key signaling receptors in collagen-dependent platelet aggregation and in arterial thrombus formation under shear. The multiple downstream signaling pathways are still poorly understood. Here, we focused on disclosing the integrin-dependent roles of focal adhesion kinase (protein tyrosine kinase 2, PTK2), the shear-dependent collagen receptor GPR56 (ADGRG1 gene), and calcium and integrin-binding protein 1 (CIB1). We designed and synthetized peptides that interfered with integrin αIIb binding (pCIB and pCIBm) or mimicked the activation of GPR56 (pGRP). The results show that the combination of pGRP with PTK2 inhibition or of pGRP with pCIB > pCIBm in additive ways suppressed collagen- and GPVI-dependent platelet activation, thrombus buildup, and contraction. Microscopic thrombus formation was assessed by eight parameters (with script descriptions enclosed). The suppressive rather than activating effects of pGRP were confined to blood flow at a high shear rate. Blockage of PTK2 or interference of CIB1 no more than slightly affected thrombus formation at a low shear rate. Peptides did not influence GPVI-induced aggregation and Ca2+ signaling in the absence of shear. Together, these data reveal a shear-dependent signaling axis of PTK2, integrin αIIbβ3, and CIB1 in collagen- and GPVI-dependent thrombus formation, which is modulated by GPR56 and exclusively at high shear. This work thereby supports the role of PTK2 in integrin αIIbβ3 activation and signaling.
Keywords: GPR56; focal adhesion kinase; integrins; platelets; thrombus formation.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures







Similar articles
-
Principal role of glycoprotein VI in alpha2beta1 and alphaIIbbeta3 activation during collagen-induced thrombus formation.Arterioscler Thromb Vasc Biol. 2004 Sep;24(9):1727-33. doi: 10.1161/01.ATV.0000137974.85068.93. Epub 2004 Jul 1. Arterioscler Thromb Vasc Biol. 2004. PMID: 15231520
-
Differential integrin activity mediated by platelet collagen receptor engagement under flow conditions.Thromb Haemost. 2017 Jul 26;117(8):1588-1600. doi: 10.1160/TH16-12-0906. Epub 2017 May 24. Thromb Haemost. 2017. PMID: 28536721 Free PMC article.
-
Platelet collagen receptor Glycoprotein VI-dimer recognizes fibrinogen and fibrin through their D-domains, contributing to platelet adhesion and activation during thrombus formation.J Thromb Haemost. 2018 Feb;16(2):389-404. doi: 10.1111/jth.13919. Epub 2018 Jan 15. J Thromb Haemost. 2018. PMID: 29210180 Free PMC article.
-
GPVI and integrin alphaIIb beta3 signaling in platelets.J Thromb Haemost. 2005 Aug;3(8):1752-62. doi: 10.1111/j.1538-7836.2005.01429.x. J Thromb Haemost. 2005. PMID: 16102042 Review.
-
Platelet physiology and thrombosis.Thromb Res. 2004;114(5-6):447-53. doi: 10.1016/j.thromres.2004.07.020. Thromb Res. 2004. PMID: 15507277 Review.
Cited by
-
Unraveling the therapeutic potential of Senna singueana phytochemicals to attenuate pancreatic cancer using protein-protein interactions, molecular docking, and MD simulation.In Silico Pharmacol. 2023 Dec 13;12(1):3. doi: 10.1007/s40203-023-00179-9. eCollection 2024. In Silico Pharmacol. 2023. PMID: 38108042 Free PMC article.
-
Structural bioinformatics for rational drug design.Res Pract Thromb Haemost. 2025 Jan 23;9(1):102691. doi: 10.1016/j.rpth.2025.102691. eCollection 2025 Jan. Res Pract Thromb Haemost. 2025. PMID: 40027444 Free PMC article.
References
-
- Nagy M., Perrella G., Dalby A., Becerra M., Garcia Quintanilla L., Pike J., Morgan N., Gardiner E., Heemskerk J.W., Azocar L., et al. Flow studies on human GPVI-deficient blood under coagulating and non-coagulating conditions. Blood Adv. 2020;4:2953–2961. doi: 10.1182/bloodadvances.2020001761. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
- 766118/European Union's Horizon 2020 research and innovation program under the Marie Sklodowska-Curie grant agreement TAPAS
- PID2019-108727RB-I00/Spanish Ministry of Science and Innovation
- 2019-2022/Sociedad Española de Trombosis y Hemostasia (SETH), and the Consellería de Cultura, Educación e Ordenación Universitaria, Xunta de Galicia (Centro Singular de investigación de Galicia ac-creditation 2019-2022).
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

