Myoglobin-Pyruvate Interactions: Binding Thermodynamics, Structure-Function Relationships, and Impact on Oxygen Release Kinetics
- PMID: 35955898
- PMCID: PMC9369265
- DOI: 10.3390/ijms23158766
Myoglobin-Pyruvate Interactions: Binding Thermodynamics, Structure-Function Relationships, and Impact on Oxygen Release Kinetics
Abstract
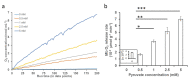
Myoglobin (Mb), besides its roles as an oxygen (O2) carrier/storage protein and nitric oxide NO scavenger/producer, may participate in lipid trafficking and metabolite binding. Our recent findings have shown that O2 is released from oxy-Mb upon interaction with lactate (LAC, anerobic glycolysis end-product). Since pyruvate (PYR) is structurally similar and metabolically related to LAC, we investigated the effects of PYR (aerobic glycolysis end-product) on Mb using isothermal titration calorimetry, circular dichroism, and O2-kinetic studies to evaluate PYR affinity toward Mb and to compare the effects of PYR and LAC on O2 release kinetics of oxy-Mb. Similar to LAC, PYR interacts with both oxy- and deoxy-Mb with a 1:1 stoichiometry. Time-resolved circular dichroism spectra revealed that there are no major conformational changes in the secondary structures of oxy- or deoxy-Mb during interactions with PYR or LAC. However, we found contrasting results with respect to binding affinities and substrate preference, where PYR has higher affinity toward deoxy-Mb when compared with LAC (which prefers oxy-Mb). Furthermore, PYR interaction with oxy-Mb releases a significantly lower amount of O2 than LAC. Taken together, our findings support the hypothesis that glycolytic end-products play a distinctive role in the Mb-rich tissues by serving as novel regulators of O2 availability, and/or by impacting other activities related to oxy-/deoxy-Mb toggling in resting vs. exercised or metabolically activated conditions.
Keywords: myoglobin; oxygen release; pyruvate.
Conflict of interest statement
The authors declare no potential conflict of interest with respect to the research, authorship, and/or publication of this article. S.H.A. is the founder and principal of XenoMed, LLC, which is focused on research and discovery that has no connection to the current project. XenoMed had no part in the research design, funding, results, or writing of the manuscript.
Figures
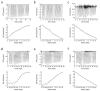
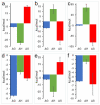
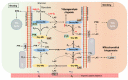
 ) display high affinity and rapid reaction rates and dashed arrows (
) display high affinity and rapid reaction rates and dashed arrows ( ) display lower affinity and slower reaction rates. oxy-Mb: oxygenated-Mb; deoxy-Mb: deoxygenated-Mb; PYR: pyruvate; LAC: lactate; TCA cycle: tricarboxylic acid cycle; ETC: electron transport chain; MPC: mitochondrial pyruvate complex; mLOC: mitochondrial lactate oxidation complex; MCT4: monocarboxylate transporter 4.
) display lower affinity and slower reaction rates. oxy-Mb: oxygenated-Mb; deoxy-Mb: deoxygenated-Mb; PYR: pyruvate; LAC: lactate; TCA cycle: tricarboxylic acid cycle; ETC: electron transport chain; MPC: mitochondrial pyruvate complex; mLOC: mitochondrial lactate oxidation complex; MCT4: monocarboxylate transporter 4.Similar articles
-
A versatile delivery vehicle for cellular oxygen and fuels or metabolic sensor? A review and perspective on the functions of myoglobin.Physiol Rev. 2024 Oct 1;104(4):1611-1642. doi: 10.1152/physrev.00031.2023. Epub 2024 May 2. Physiol Rev. 2024. PMID: 38696337 Free PMC article. Review.
-
Myoglobin Interaction with Lactate Rapidly Releases Oxygen: Studies on Binding Thermodynamics, Spectroscopy, and Oxygen Kinetics.Int J Mol Sci. 2022 Apr 26;23(9):4747. doi: 10.3390/ijms23094747. Int J Mol Sci. 2022. PMID: 35563138 Free PMC article.
-
Novel Molecular Interactions of Acylcarnitines and Fatty Acids with Myoglobin.J Biol Chem. 2016 Nov 25;291(48):25133-25143. doi: 10.1074/jbc.M116.754978. Epub 2016 Oct 7. J Biol Chem. 2016. PMID: 27758871 Free PMC article.
-
Specific modification of structure and property of myoglobin by the formation of tetrazolylhistidine 64(E7). Reaction of the modified myoglobin with molecular oxygen.J Biol Chem. 1993 Sep 25;268(27):19983-90. J Biol Chem. 1993. PMID: 8397193
-
Myoglobin: Oxygen Depot or Oxygen Transporter to Mitochondria? A Novel Mechanism of Myoglobin Deoxygenation in Cells (review).Biochemistry (Mosc). 2018 Feb;83(2):168-183. doi: 10.1134/S0006297918020098. Biochemistry (Mosc). 2018. PMID: 29618303 Review.
Cited by
-
Computational Analysis Reveals Unique Binding Patterns of Oxygenated and Deoxygenated Myoglobin to the Outer Mitochondrial Membrane.Biomolecules. 2023 Jul 17;13(7):1138. doi: 10.3390/biom13071138. Biomolecules. 2023. PMID: 37509174 Free PMC article.
-
Unraveling Protein-Metabolite Interactions in Precision Nutrition: A Case Study of Blueberry-Derived Metabolites Using Advanced Computational Methods.Metabolites. 2024 Aug 3;14(8):430. doi: 10.3390/metabo14080430. Metabolites. 2024. PMID: 39195526 Free PMC article.
-
A versatile delivery vehicle for cellular oxygen and fuels or metabolic sensor? A review and perspective on the functions of myoglobin.Physiol Rev. 2024 Oct 1;104(4):1611-1642. doi: 10.1152/physrev.00031.2023. Epub 2024 May 2. Physiol Rev. 2024. PMID: 38696337 Free PMC article. Review.
-
Glycogen Supplementation in Vitro Promotes pH Decline in Dark-Cutting Beef by Reverting Muscle's Metabolome toward a Normal Postmortem Muscle State.J Agric Food Chem. 2024 Nov 13;72(45):25275-25285. doi: 10.1021/acs.jafc.4c06490. Epub 2024 Nov 4. J Agric Food Chem. 2024. PMID: 39496138 Free PMC article.
References
-
- Voet D., Voet J.G., Pratt C.W. Fundamentals of Biochemistry Life at the Molecular Level. Wiley; Hoboken, NJ, USA: 2016.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

