Protective Effects of Recombinant Human Angiogenin in Keratinocytes: New Insights on Oxidative Stress Response Mediated by RNases
- PMID: 35955913
- PMCID: PMC9369303
- DOI: 10.3390/ijms23158781
Protective Effects of Recombinant Human Angiogenin in Keratinocytes: New Insights on Oxidative Stress Response Mediated by RNases
Abstract
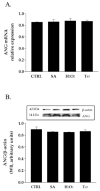
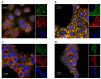
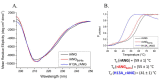
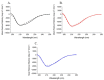
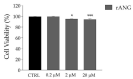
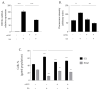
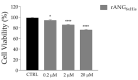
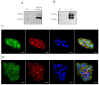
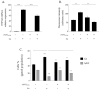
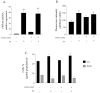
Human angiogenin (ANG) is a 14-kDa ribonuclease involved in different pathophysiological processes including tumorigenesis, neuroprotection, inflammation, innate immunity, reproduction, the regeneration of damaged tissues and stress cell response, depending on its intracellular localization. Under physiological conditions, ANG moves to the cell nucleus where it enhances rRNA transcription; conversely, recent reports indicate that under stress conditions, ANG accumulates in the cytoplasmic compartment and modulates the production of tiRNAs, a novel class of small RNAs that contribute to the translational inhibition and recruitment of stress granules (SGs). To date, there is still limited and controversial experimental evidence relating to a hypothetical role of ANG in the epidermis, the outermost layer of human skin, which is continually exposed to external stressors. The present study collects compelling evidence that endogenous ANG is able to modify its subcellular localization on HaCaT cells, depending on different cellular stresses. Furthermore, the use of recombinant ANG allowed to determine as this special enzyme is effectively able to counter at various levels the alterations of cellular homeostasis in HaCaT cells, actually opening a new vision on the possible functions that this special enzyme can support also in the stress response of human skin.
Keywords: oxidative stress; skin cells; stress granules; stress induced RNases; tiRNA.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
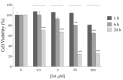

Similar articles
-
In lysate RNA digestion provides insights into the angiogenin's specificity towards transfer RNAs.RNA Biol. 2021 Dec;18(12):2546-2555. doi: 10.1080/15476286.2021.1930758. Epub 2021 Jun 4. RNA Biol. 2021. PMID: 34085908 Free PMC article.
-
Ribonuclease/angiogenin inhibitor 1 regulates stress-induced subcellular localization of angiogenin to control growth and survival.J Cell Sci. 2013 Sep 15;126(Pt 18):4308-19. doi: 10.1242/jcs.134551. Epub 2013 Jul 10. J Cell Sci. 2013. PMID: 23843625 Free PMC article.
-
Emerging role of angiogenin in stress response and cell survival under adverse conditions.J Cell Physiol. 2012 Jul;227(7):2822-6. doi: 10.1002/jcp.23051. J Cell Physiol. 2012. PMID: 22021078 Free PMC article. Review.
-
Angiogenin Prevents Progranulin A9D Mutation-Induced Neuronal-Like Cell Apoptosis Through Cleaving tRNAs into tiRNAs.Mol Neurobiol. 2018 Feb;55(2):1338-1351. doi: 10.1007/s12035-017-0396-7. Epub 2017 Jan 27. Mol Neurobiol. 2018. PMID: 28127696
-
RNA biology of angiogenin: Current state and perspectives.RNA Biol. 2017 Feb;14(2):171-178. doi: 10.1080/15476286.2016.1272746. RNA Biol. 2017. PMID: 28010172 Free PMC article. Review.
Cited by
-
Study of the Antimicrobial Activity of the Human Peptide SQQ30 against Pathogenic Bacteria.Antibiotics (Basel). 2024 Feb 1;13(2):145. doi: 10.3390/antibiotics13020145. Antibiotics (Basel). 2024. PMID: 38391531 Free PMC article.
-
Assembly and disassembly of stress granules in kidney diseases.iScience. 2025 May 24;28(6):112578. doi: 10.1016/j.isci.2025.112578. eCollection 2025 Jun 20. iScience. 2025. PMID: 40546967 Free PMC article. Review.
-
Simultaneous Irradiation with UV-A, -B, and -C Lights Promotes Effective Decontamination of Planktonic and Sessile Bacteria: A Pilot Study.Int J Mol Sci. 2023 Aug 18;24(16):12951. doi: 10.3390/ijms241612951. Int J Mol Sci. 2023. PMID: 37629131 Free PMC article.
-
Biotechnological Potential of a Novel Strain of Fusarium proliferatum, a Terrestrial Fungus Adapted to Marine Environment.Environ Microbiol Rep. 2025 Aug;17(4):e70143. doi: 10.1111/1758-2229.70143. Environ Microbiol Rep. 2025. PMID: 40611708 Free PMC article.
-
Effects of Pathogenic Mutants of the Neuroprotective RNase 5-Angiogenin in Amyotrophic Lateral Sclerosis (ALS).Genes (Basel). 2024 Jun 4;15(6):738. doi: 10.3390/genes15060738. Genes (Basel). 2024. PMID: 38927674 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

