Downregulation of the Cd38-Cyclic ADP-Ribose Signaling in Cardiomyocytes by Intermittent Hypoxia via Pten Upregulation
- PMID: 35955916
- PMCID: PMC9368863
- DOI: 10.3390/ijms23158782
Downregulation of the Cd38-Cyclic ADP-Ribose Signaling in Cardiomyocytes by Intermittent Hypoxia via Pten Upregulation
Abstract
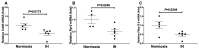
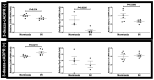
Sleep apnea syndrome (SAS) is characterized by recurrent episodes of oxygen desaturation and reoxygenation (intermittent hypoxia, IH), and it is a risk factor for cardiovascular disease (CVD) and insulin resistance/type 2 diabetes. However, the mechanisms linking IH stress and CVD remain elusive. We exposed rat H9c2 and mouse P19.CL6 cardiomyocytes to experimental IH or normoxia for 24 h to analyze the mRNA expression of the components of Cd38-cyclic ADP-ribose (cADPR) signaling. We found that the mRNA levels of cluster of differentiation 38 (Cd38), type 2 ryanodine receptor (Ryr2), and FK506-binding protein 12.6 (Fkbp12.6) in H9c2 and P19.CL6 cardiomyocytes were significantly decreased by IH, whereas the promoter activities of these genes were not decreased. By contrast, the expression of phosphatase and tensin homolog deleted from chromosome 10 (Pten) was upregulated in IH-treated cells. The small interfering RNA for Pten (siPten) and a non-specific control RNA were introduced into the H9c2 cells. The IH-induced downregulation of Cd38, Ryr2, and Fkbp12.6 was abolished by the introduction of the siPten, but not by the control RNA. These results indicate that IH stress upregulated the Pten in cardiomyocytes, resulting in the decreased mRNA levels of Cd38, Ryr2, and Fkbp12.6, leading to the inhibition of cardiomyocyte functions in SAS patients.
Keywords: Cd38; Fkbp12.6; Pten; Ryr2; cardiomyocytes; intermittent hypoxia; sleep apnea syndrome.
Conflict of interest statement
All authors state that they have no conflicts of interest.
Figures


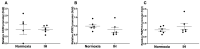



Similar articles
-
Upregulation of Reg IV and Hgf mRNAs by Intermittent Hypoxia via Downregulation of microRNA-499 in Cardiomyocytes.Int J Mol Sci. 2022 Oct 17;23(20):12414. doi: 10.3390/ijms232012414. Int J Mol Sci. 2022. PMID: 36293268 Free PMC article.
-
Intermittent Hypoxia Upregulates the Renin and Cd38 mRNAs in Renin-Producing Cells via the Downregulation of miR-203.Int J Mol Sci. 2021 Sep 19;22(18):10127. doi: 10.3390/ijms221810127. Int J Mol Sci. 2021. PMID: 34576290 Free PMC article.
-
Attenuation of glucose-induced insulin secretion by intermittent hypoxia via down-regulation of CD38.Life Sci. 2012 Jan 30;90(5-6):206-11. doi: 10.1016/j.lfs.2011.11.011. Epub 2011 Dec 1. Life Sci. 2012. PMID: 22154909
-
CD38-Cyclic ADP-Ribose Signal System in Physiology, Biochemistry, and Pathophysiology.Int J Mol Sci. 2022 Apr 13;23(8):4306. doi: 10.3390/ijms23084306. Int J Mol Sci. 2022. PMID: 35457121 Free PMC article. Review.
-
["The CD38-cyclic ADP-ribose signal system": molecular mechanism and biological significance].Nihon Yakurigaku Zasshi. 1999 Sep;114(3):131-9. doi: 10.1254/fpj.114.131. Nihon Yakurigaku Zasshi. 1999. PMID: 10553576 Review. Japanese.
Cited by
-
Chronic intermittent hypoxia aggravated diabetic cardiomyopathy through LKB1/AMPK/Nrf2 signaling pathway.PLoS One. 2024 Mar 7;19(3):e0296792. doi: 10.1371/journal.pone.0296792. eCollection 2024. PLoS One. 2024. PMID: 38452099 Free PMC article.
-
The Impact of Intermittent Hypoxia on Metabolism and Cognition.Int J Mol Sci. 2022 Oct 26;23(21):12957. doi: 10.3390/ijms232112957. Int J Mol Sci. 2022. PMID: 36361741 Free PMC article. Review.
-
Upregulation of Reg IV and Hgf mRNAs by Intermittent Hypoxia via Downregulation of microRNA-499 in Cardiomyocytes.Int J Mol Sci. 2022 Oct 17;23(20):12414. doi: 10.3390/ijms232012414. Int J Mol Sci. 2022. PMID: 36293268 Free PMC article.
-
Possible Molecular Mechanisms of Hypertension Induced by Sleep Apnea Syndrome/Intermittent Hypoxia.Life (Basel). 2024 Jan 22;14(1):157. doi: 10.3390/life14010157. Life (Basel). 2024. PMID: 38276286 Free PMC article. Review.
-
CD38 contributes to tumor progression and tumor microenvironment reshaping in epithelial ovarian cancer.Transl Oncol. 2025 Jul;57:102414. doi: 10.1016/j.tranon.2025.102414. Epub 2025 May 16. Transl Oncol. 2025. PMID: 40381484 Free PMC article.
References
-
- Ota H., Takasawa S., Yamauchi M., Yoshikawa M., Tomoda K., Kimura H. Intermittent hypoxia in pancreatic beta cells. Pancreat. Disord. Ther. 2015;5:S5-004. doi: 10.4172/2165-7092.S5-004. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

