Anticancer Effect of Cathelicidin LL-37, Protegrin PG-1, Nerve Growth Factor NGF, and Temozolomide: Impact on the Mitochondrial Metabolism, Clonogenic Potential, and Migration of Human U251 Glioma Cells
- PMID: 35956937
- PMCID: PMC9370145
- DOI: 10.3390/molecules27154988
Anticancer Effect of Cathelicidin LL-37, Protegrin PG-1, Nerve Growth Factor NGF, and Temozolomide: Impact on the Mitochondrial Metabolism, Clonogenic Potential, and Migration of Human U251 Glioma Cells
Abstract
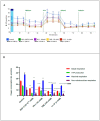
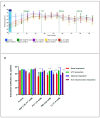
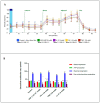
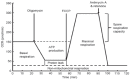
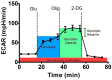
Glioblastoma (GBM) is one of the most aggressive and lethal malignancy of the central nervous system. Temozolomide is the standard of care for gliomas, frequently results in resistance to drug and tumor recurrence. Therefore, further research is required for the development of effective drugs in order to guarantee specific treatments to succeed. The aim of current study was to investigate the effects of nerve growth factor (NGF), human cathelicidin (LL-37), protegrin-1 (PG-1), and temozolomide on bioenergetic function of mitochondria, clonogenicity, and migration of human U251 glioma cells. Colony formation assay was used to test the ability of the glioma cells to form colonies in vitro. The U251 glioma cells migration was evaluated using wound-healing assay. To study the mitochondrial metabolism in glioma cells we measured oxygen consumption rates (OCR) and extracellular acidification rates (ECAR) using a Seahorse XF cell Mito stress test kit and Seahorse XF cell Glycolysis stress kit, respectively. We revealed that LL-37, NGF, and TMZ show strong anti-tumorigenic activity on GMB. LL-37 (4 μM), TMZ (155 μM), and NGF (7.55 × 10-3 μM) inhibited 43.9%-60.3%, 73.5%-81.3%, 66.2% the clonogenicity of glioma U251 cells for 1-2 days, respectively. LL-37 (4 μM), and NGF (7.55 × 10-3 μM) inhibited the migration of U251 glioma cells on the third and fourth days. TMZ also inhibited the migration of human glioma U251 cells over 1-3 days. In contrast, PG-1 (16 μM) stimulated the migration of U251 glioma cells on the second, fourth, and sixth days. Anti-mitogenic and anti-migration activities of NGF, LL-37, and TMZ maybe are relation to their capacity to reduce the basal OCR, ATP-synthetase, and maximal respiration of mitochondria in human glioma U251 cells. Glycolysis, glycolytic capacity and glycolytic spare in glioma U251 cells haven`t been changed under the effect of NGF, LL-37, PG-1, and TMZ in regard to control level. Thus, LL-37 and NGF inhibit migration and clonogenicity of U251 glioma cells, which may indicate that these compounds have anti-mitogenic and anti-migration effects on human glioma cells. The study of the mechanisms of these effects may contribute in the future to the use of NGF and LL-37 as therapeutic agents for gliomas.
Keywords: ECAR; OCR; cathelicidin LL-37; clonogenicity; human glioma U251; metabolism of mitochondria; migration; nerve growth factor NGF; protegrin PG-1; temozolomide.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The authors declare no conflict of interest.
Figures


References
-
- International Agency for Research of Cancer (Globocan) [Electronic Resource]: The World of Health Organization. 2020. [(accessed on 23 December 2021)]. Available online: www.globocan.iarc.fr.
-
- Skaga E., Kulesskiy E., Fayzullin A., Sandberg C.J., Potdar S., Kyttälä A., Langmoen I.A., Laakso A., Gaál-Paavola E., Perola M., et al. Intertumoral heterogeneity in patient-specific drug sensitivities in treatment-naïve glioblastoma. BMC Cancer. 2019;19:628. doi: 10.1186/s12885-019-5861-4. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

