The Relationship between Procyanidin Structure and Their Protective Effect in a Parkinson's Disease Model
- PMID: 35956957
- PMCID: PMC9370466
- DOI: 10.3390/molecules27155007
The Relationship between Procyanidin Structure and Their Protective Effect in a Parkinson's Disease Model
Abstract
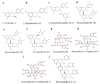
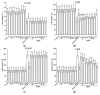
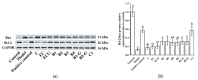
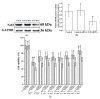
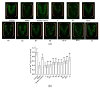
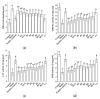
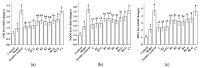
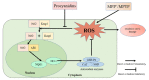
This study evaluated the effect of grape seed-derived monomer, dimeric, and trimeric procyanidins on rat pheochromocytoma cell line (PC12) cells and in a zebrafish Parkinson's disease (PD) model. PC12 cells were cultured with grape seed-derived procyanidins or deprenyl for 24 h and then exposed to 1.5 mm 1-methyl-4-phenylpyridinium (MPP+) for 24 h. Zebrafish larvae (AB strain) 3 days post-fertilization were incubated with deprenyl or grape seed-derived procyanidins in 400 µM 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) for 4 days. The results showed that the procyanidin dimers procyanidin B1 (B1), procyanidin B2 (B2), procyanidin B3 (B3), procyanidin B4 (B4), procyanidin B1-3-O-gallate (B1-G), procyanidin B2-3-O-gallate (B2-G), and the procyanidin trimer procyanidin C1 (C1) had a protective effect on PC12 cells, decreasing the damaged dopaminergic neurons and motor impairment in zebrafish. In PC12 cells and the zebrafish PD model, procyanidin (B1, B2, B3, B4, B1-G, B2-G, C1) treatment decreased the content of reactive oxygen species (ROS) and malondialdehyde (MDA), increased the activity of antioxidant enzymes glutathione peroxidase (GSH-Px), catalase (CAT), and superoxide dismutase (SOD), and upregulated the expression of nuclear factor-erythroid 2-related factor (Nrf2), NAD(P)H: quinone oxidoreductase 1 (NQO1), and heme oxygenase-1 (HO-1). These results suggest that in PC12 cells and the zebrafish PD model, the neuroprotective effects of the procyanidins were positively correlated with their degree of polymerization.
Keywords: Nrf2/ARE pathway; PC12 cells; Parkinson’s disease; procyanidins; zebrafish.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Relationship between Neuroprotective Effects and Structure of Procyanidins.Molecules. 2022 Apr 2;27(7):2308. doi: 10.3390/molecules27072308. Molecules. 2022. PMID: 35408708 Free PMC article.
-
Protective Effects and Mechanisms of Procyanidins on Parkinson's Disease In Vivo and In Vitro.Molecules. 2021 Sep 13;26(18):5558. doi: 10.3390/molecules26185558. Molecules. 2021. PMID: 34577027 Free PMC article.
-
Neuroprotective Effects and Mechanisms of Procyanidins In Vitro and In Vivo.Molecules. 2021 May 17;26(10):2963. doi: 10.3390/molecules26102963. Molecules. 2021. PMID: 34067571 Free PMC article.
-
Antioxidant mechanism and protection of nigral neurons against MPP+ toxicity by deprenyl (selegiline).Ann N Y Acad Sci. 1994 Nov 17;738:214-21. doi: 10.1111/j.1749-6632.1994.tb21806.x. Ann N Y Acad Sci. 1994. PMID: 7832430 Review.
-
Autophagy as an essential cellular antioxidant pathway in neurodegenerative disease.Redox Biol. 2013 Dec 25;2:82-90. doi: 10.1016/j.redox.2013.12.013. eCollection 2014. Redox Biol. 2013. PMID: 24494187 Free PMC article. Review.
Cited by
-
Potential Roles of Natural Antioxidants in Modulating Neurodegenerative Disease Pathways.Mol Neurobiol. 2025 Aug;62(8):10367-10397. doi: 10.1007/s12035-025-04874-w. Epub 2025 Apr 9. Mol Neurobiol. 2025. PMID: 40202704 Review.
-
Rust Conversion of Proanthocyanidins to Archaeological Steel: A Case Study of Lingzhao Xuan in the Forbidden City.Molecules. 2022 Nov 9;27(22):7711. doi: 10.3390/molecules27227711. Molecules. 2022. PMID: 36431813 Free PMC article.
References
-
- Schapira A.H.V. Pathogenesis of Parkinson’s disease. Bailliere Clin. Neur. 1997;6:15–36. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

