Electro-Acupuncture Regulates Metabolic Disorders of the Liver and Kidney in Premature Ovarian Failure Mice
- PMID: 35957829
- PMCID: PMC9359440
- DOI: 10.3389/fendo.2022.882214
Electro-Acupuncture Regulates Metabolic Disorders of the Liver and Kidney in Premature Ovarian Failure Mice
Abstract
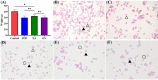
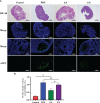
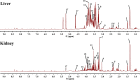
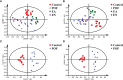
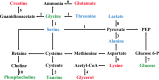
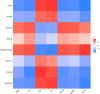
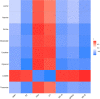
As per the theory of traditional Chinese medicine (TCM), the liver and kidney dysfunction are important pathogenies for premature ovarian failure (POF). POF is a common gynecological disease that reduced the pregnancy rate. Electro-acupuncture (EA) is a useful non-pharmaceutical therapy that supposedly regulates the function of the liver and kidney in the treatment of POF with TCM. However, the underlying mechanism of EA in the treatment of POF has not been adequately studied through metabonomics with reference to the theory of TCM. Accordingly, we investigated the effect of EA on the liver and kidney metabolites in POF mice through metabolomics. POF mice were established via intraperitoneal injection of cisplatin. Both Sanyinjiao (SP6) and Guanyuan (CV4) were stimulated by EA for 3 weeks. The biological samples (including the serum and the ovary, liver, and kidney tissues) were evaluated by histopathology, molecular biology, and hydrogen-1 nuclear magnetic resonance (1HNMR)-based metabolomics to assess the efficacy of EA. 1HNMR data were analyzed by the orthogonal partial least squares discriminant analysis (OPLS-DA). The results revealed that EA was beneficial to ovarian function and the menstrual cycle of POF. Both the energy metabolism and neurotransmitter metabolism in the liver and kidney were regulated by EA. Notably, EA played an important role in regulating energy-related metabolism in the kidney, and the better effect of neurotransmitter-related metabolism in the liver was regulated by EA. These findings indicated that the ovarian functions could be improved and the metabolic disorder of the liver and kidney caused by POF could be regulated by EA. Our study results thus suggested that the EA therapy, based on the results for the liver and kidney, were related to POF in TCM, as preliminarily confirmed through metabolomics.
Keywords: 1HNMR; electro-acupuncture; energy metabolism; metabonomics; neurotransmitter metabolism; premature ovarian failure.
Copyright © 2022 Chen, He, Guo, Wu, Zhang, Yau, Xie, Guo, Tong, Yang and Xiao.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





Similar articles
-
[Pseudotargeted metabolomics analysis of pine pollen intervention in the liver of premature ovarian failure rats].Se Pu. 2023 Jan;41(1):47-57. doi: 10.3724/SP.J.1123.2022.04017. Se Pu. 2023. PMID: 36633076 Free PMC article. Chinese.
-
[Clinical research and the effect mechanism on premature ovarian failure treated with acupuncture in recent 20 years].Zhongguo Zhen Jiu. 2018 May 12;38(5):5653-70. doi: 10.13703/j.0255-2930.2018.05.031. Zhongguo Zhen Jiu. 2018. PMID: 29797923 Review. Chinese.
-
Electro-acupuncture attenuates the mice premature ovarian failure via mediating PI3K/AKT/mTOR pathway.Life Sci. 2019 Jan 15;217:169-175. doi: 10.1016/j.lfs.2018.11.059. Epub 2018 Dec 3. Life Sci. 2019. PMID: 30521869
-
Effects of Electroacupuncture on the Gut Microbiome in Cisplatin-Induced Premature Ovarian Failure Mice.Evid Based Complement Alternat Med. 2022 Mar 14;2022:9352833. doi: 10.1155/2022/9352833. eCollection 2022. Evid Based Complement Alternat Med. 2022. PMID: 35321505 Free PMC article.
-
[Mechanism of tonifying kidney and activating blood therapy for premature ovarian failure:a review].Zhongguo Zhong Yao Za Zhi. 2023 Apr;48(7):1808-1814. doi: 10.19540/j.cnki.cjcmm.20230105.501. Zhongguo Zhong Yao Za Zhi. 2023. PMID: 37282955 Review. Chinese.
Cited by
-
MicroRNA-22-3p in human umbilical cord mesenchymal stem cell-secreted exosomes inhibits granulosa cell apoptosis by targeting KLF6 and ATF4-ATF3-CHOP pathway in POF mice.Apoptosis. 2023 Aug;28(7-8):997-1011. doi: 10.1007/s10495-023-01833-5. Epub 2023 Mar 31. Apoptosis. 2023. PMID: 37000316
-
Mechanisms underlying acupuncture therapy in chronic kidney disease: A narrative overview of preclinical studies and clinical trials.Front Nephrol. 2022 Nov 9;2:1006506. doi: 10.3389/fneph.2022.1006506. eCollection 2022. Front Nephrol. 2022. PMID: 37675019 Free PMC article. Review.
-
The quality and reliability of short videos about premature ovarian failure on Bilibili and TikTok: Cross-sectional study.Digit Health. 2025 Jun 17;11:20552076251351077. doi: 10.1177/20552076251351077. eCollection 2025 Jan-Dec. Digit Health. 2025. PMID: 40534890 Free PMC article.
-
Electroacupuncture enhances the mitophagy of granulosa cells in premature ovarian insufficiency model mice by inactivating the hippo-yes-associated protein/transcriptional co-activator with postsynaptic density protein, drosophila disc large tumor suppressor, and zonula occludens-1 protein binding motif pathway.J Tradit Chin Med. 2025 Feb;45(1):13-21. doi: 10.19852/j.cnki.jtcm.2025.01.002. J Tradit Chin Med. 2025. PMID: 39957154 Free PMC article.
-
Acupuncture attenuates experimental autoimmune thyroiditis by modulating intestinal microbiota and palmitic acid metabolism.Front Immunol. 2025 Apr 28;16:1541728. doi: 10.3389/fimmu.2025.1541728. eCollection 2025. Front Immunol. 2025. PMID: 40356922 Free PMC article.
References
-
- Igboeli P, El Andaloussi A, Sheikh U, Takala H, ElSharoud A, McHugh A, et al. . Intraovarian Injection of Autologous Human Mesenchymal Stem Cells Increases Estrogen Production and Reduces Menopausal Symptoms in Women With Premature Ovarian Failure: Two Case Reports and a Review of the Literature. J Med Case Rep (2020) 14(1):108. doi: 10.1186/s13256-020-02426-5 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

