Efficacy of SARS-CoV-2 vaccination in patients with monoclonal gammopathies: A cross sectional study
- PMID: 35961779
- PMCID: PMC9375155
- DOI: 10.26508/lsa.202201479
Efficacy of SARS-CoV-2 vaccination in patients with monoclonal gammopathies: A cross sectional study
Abstract
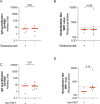
SARS-CoV-2 vaccination is the most effective strategy to protect individuals with haematologic malignancies against severe COVID-19, while eliciting limited vaccine responses. We characterized the humoral responses following 3 mo after mRNA-based vaccines in individuals at different plasma-cell disease stages: monoclonal gammopathy of undetermined significance (MGUS), smoldering multiple myeloma (SMM), and multiple myeloma on first-line therapy (MM), compared with a healthy population. Plasma samples from uninfected MM patients showed lower SARS-CoV-2-specific antibody levels and neutralization capacity compared with MGUS, SMM, and healthy individuals. Importantly, COVID-19 recovered MM individuals presented significantly higher plasma neutralization capacity compared with their uninfected counterparts, highlighting that hybrid immunity elicit stronger immunity even in this immunocompromised population. No differences in the vaccine-induced humoral responses were observed between uninfected MGUS, SMM and healthy individuals. In conclusion, MGUS and SMM patients could be SARS-CoV-2 vaccinated following the vaccine recommendations for the general population, whereas a tailored monitoring of the vaccine-induced immune responses should be considered in uninfected MM patients.
© 2022 Abella et al.
Conflict of interest statement
J Blanco and J Carrillo reports personal fees from Albajuna Therapeutics, outside the submitted work.
Figures




References
-
- Chung DJ, Shah GL, Devlin SM, Ramanathan LV, Doddi S, Pessin MS, Hoover E, Marcello LT, Young JC, Boutemine SR, et al. (2021) Disease- and therapy-specific impact on humoral immune responses to COVID-19 vaccination in hematologic malignancies. Blood Cancer Discov 2: 568–576. 10.1158/2643-3230.bcd-21-0139 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
