Somatodendritic Release of Cholecystokinin Potentiates GABAergic Synapses Onto Ventral Tegmental Area Dopamine Cells
- PMID: 35961792
- PMCID: PMC9976994
- DOI: 10.1016/j.biopsych.2022.06.011
Somatodendritic Release of Cholecystokinin Potentiates GABAergic Synapses Onto Ventral Tegmental Area Dopamine Cells
Abstract
Background: Neuropeptides are contained in nearly every neuron in the central nervous system and can be released not only from nerve terminals but also from somatodendritic sites. Cholecystokinin (CCK), among the most abundant neuropeptides in the brain, is expressed in the majority of midbrain dopamine neurons. Despite this high expression, CCK function within the ventral tegmental area (VTA) is not well understood.
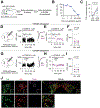
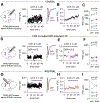
Methods: We confirmed CCK expression in VTA dopamine neurons through immunohistochemistry and in situ hybridization and detected optogenetically induced CCK release using an enzyme-linked immunosorbent assay. To investigate whether CCK modulates VTA circuit activity, we used whole-cell patch clamp recordings in mouse brain slices. We infused CCK locally in vivo and tested food intake and locomotion in fasted mice. We also used in vivo fiber photometry to measure Ca2+ transients in dopamine neurons during feeding.
Results: Here we report that VTA dopamine neurons release CCK from somatodendritic regions, where it triggers long-term potentiation of GABAergic (gamma-aminobutyric acidergic) synapses. The somatodendritic release occurs during trains of optogenetic stimuli or prolonged but modest depolarization and is dependent on synaptotagmin-7 and T-type Ca2+ channels. Depolarization-induced long-term potentiation is blocked by a CCK2 receptor antagonist and mimicked by exogenous CCK. Local infusion of CCK in vivo inhibits food consumption and decreases distance traveled in an open field test. Furthermore, intra-VTA-infused CCK reduced dopamine cell Ca2+ signals during food consumption after an overnight fast and was correlated with reduced food intake.
Conclusions: Our experiments introduce somatodendritic neuropeptide release as a previously unknown feedback regulator of VTA dopamine cell excitability and dopamine-related behaviors.
Keywords: Cholecystokinin; Dopamine; Feeding; Neuropeptide; Somatodendritic release; Ventral tegmental area.
Copyright © 2022 Society of Biological Psychiatry. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors report no biomedical financial interests or potential conflicts of interest.
Figures

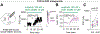


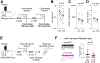
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

