Pronounced antiseizure activity of the subtype-selective GABAA positive allosteric modulator darigabat in a mouse model of drug-resistant focal epilepsy
- PMID: 35965432
- PMCID: PMC9532903
- DOI: 10.1111/cns.13927
Pronounced antiseizure activity of the subtype-selective GABAA positive allosteric modulator darigabat in a mouse model of drug-resistant focal epilepsy
Abstract
Aim: Darigabat is an α2/3/5 subunit-selective positive allosteric modulator of GABAA receptors that has demonstrated broad-spectrum activity in several preclinical models of epilepsy as well as in a clinical photoepilepsy trial. The objective here was to assess the acute antiseizure effect of darigabat in the mesial temporal lobe epilepsy (MTLE) mouse model of drug-resistant focal seizures.
Methods: The MTLE model is generated by single unilateral intrahippocampal injection of low dose (1 nmole) kainic acid in adult mice, and subsequent epileptiform activity is recorded following implantation of a bipolar electrode under general anesthesia. After a period of epileptogenesis (~4 weeks), spontaneous and recurrent hippocampal paroxysmal discharges (HPD; focal seizures) are recorded using intracerebral electroencephalography. The number and cumulated duration of HPDs were recorded following administration of vehicle (PO), darigabat (0.3-10 mg kg-1 , PO), and positive control diazepam (2 mg kg-1 , IP).
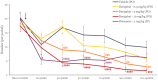
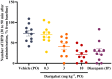
Results: Darigabat dose-dependently reduced the expression of HPDs, demonstrating comparable efficacy profile to diazepam at doses of 3 and 10 mg kg-1 .
Conclusions: Darigabat exhibited a robust efficacy profile in the MTLE model, a preclinical model of drug-resistant focal epilepsy. A Phase II proof-of-concept placebo-controlled, adjunctive-therapy trial (NCT04244175) is ongoing to evaluate efficacy and safety of darigabat in patients with drug-resistant focal seizures.
Trial registration: ClinicalTrials.gov NCT04244175 NCT04686786.
Keywords: CVL-865; GABA; MTLE; darigabat; drug-resistant epilepsy; focal; gaba; seizure (total ≥5, ≤8).
© 2022 Cerevel Therapeutics, LLC. CNS Neuroscience & Therapeutics published by John Wiley & Sons Ltd.
Conflict of interest statement
RG and PI are or were employees of Cerevel Therapeutics at the time of this research and may own stock and/or stock options in the company.
Figures

Similar articles
-
New GABA-Targeting Therapies for the Treatment of Seizures and Epilepsy: II. Treatments in Clinical Development.CNS Drugs. 2023 Sep;37(9):781-795. doi: 10.1007/s40263-023-01025-4. Epub 2023 Aug 21. CNS Drugs. 2023. PMID: 37603261 Free PMC article. Review.
-
Characterization of the intrahippocampal kainic acid model in female mice with a special focus on seizure suppression by antiseizure medications.Exp Neurol. 2024 Jun;376:114749. doi: 10.1016/j.expneurol.2024.114749. Epub 2024 Mar 11. Exp Neurol. 2024. PMID: 38467356 Free PMC article.
-
Differential Effects of Antiepileptic Drugs on Focal Seizures in the Intrahippocampal Kainate Mouse Model of Mesial Temporal Lobe Epilepsy.CNS Neurosci Ther. 2016 Jun;22(6):497-506. doi: 10.1111/cns.12523. Epub 2016 Feb 22. CNS Neurosci Ther. 2016. PMID: 26899987 Free PMC article.
-
ENX-101, a GABAA receptor α2,3,5-selective positive allosteric modulator, displays antiseizure effects in rodent seizure and epilepsy models.Epilepsia. 2025 Jun;66(6):2124-2136. doi: 10.1111/epi.18340. Epub 2025 Mar 15. Epilepsia. 2025. PMID: 40088186 Free PMC article.
-
GABAkines - Advances in the discovery, development, and commercialization of positive allosteric modulators of GABAA receptors.Pharmacol Ther. 2022 Jun;234:108035. doi: 10.1016/j.pharmthera.2021.108035. Epub 2021 Nov 16. Pharmacol Ther. 2022. PMID: 34793859 Free PMC article. Review.
Cited by
-
Genetic Background of Epilepsy and Antiepileptic Treatments.Int J Mol Sci. 2023 Nov 14;24(22):16280. doi: 10.3390/ijms242216280. Int J Mol Sci. 2023. PMID: 38003469 Free PMC article. Review.
-
Insights and progress on the biosynthesis, metabolism, and physiological functions of gamma-aminobutyric acid (GABA): a review.PeerJ. 2024 Dec 16;12:e18712. doi: 10.7717/peerj.18712. eCollection 2024. PeerJ. 2024. PMID: 39703920 Free PMC article. Review.
-
The human photosensitive epilepsy model for clinical proof-of-principle trials of novel antiseizure medications: 2. Analysis of drug trials and predictive value of the model.Epilepsia. 2025 Aug;66(8):2619-2638. doi: 10.1111/epi.18444. Epub 2025 May 10. Epilepsia. 2025. PMID: 40347110 Free PMC article. Review.
-
New epilepsy therapies in development.Nat Rev Drug Discov. 2024 Sep;23(9):682-708. doi: 10.1038/s41573-024-00981-w. Epub 2024 Jul 22. Nat Rev Drug Discov. 2024. PMID: 39039153 Review.
-
New GABA-Targeting Therapies for the Treatment of Seizures and Epilepsy: II. Treatments in Clinical Development.CNS Drugs. 2023 Sep;37(9):781-795. doi: 10.1007/s40263-023-01025-4. Epub 2023 Aug 21. CNS Drugs. 2023. PMID: 37603261 Free PMC article. Review.
References
-
- Loscher W. Fit for purpose application of currently existing animal models in the discovery of novel epilepsy therapies. Epilepsy Res. 2016;126:157‐184. - PubMed
-
- Riban V, Bouilleret V, Pham‐Le B, Fritschy J. Evolution of hippocampal epileptic activity during the development of hippocampal sclerosis in a mouse model of temporal lobe epilepsy. Neurosci. 2002;112:101‐111. - PubMed
-
- Kwan P, Arzimanoglou A, Berg AT, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc task force of the ILAE commission on therapeutic strategies. Epilepsia. 2010;51(6):1069‐1077. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

