SOX9 in Keratinocytes Regulates Claudin 2 Transcription during Skin Aging
- PMID: 35965621
- PMCID: PMC9357741
- DOI: 10.1155/2022/6884308
SOX9 in Keratinocytes Regulates Claudin 2 Transcription during Skin Aging
Abstract
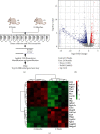
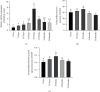
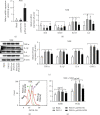
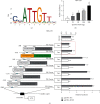
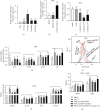
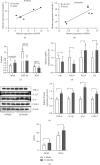
In order to prove that SOX9 in keratinocytes regulates claudin 2 transcription during skin aging, the skin of 8-week-old and 24-month-old mice is sequenced to obtain a differentially expressed gene SOX9. The gene is mainly expressed in keratinocytes, and it increases first and then decreases from newborn to aging. Six core sequences of SOX9 and claudin 2 are predicted from Jaspar. The double Luciferase Report shows that overexpression of SOX9 induces the full-length promoter of claudin 2 significantly and has no effect on the mutation and cleavage plasmid without SOX9 response. Claudin 2 is consistent with SOX9 in the skin of mice of different ages, and SOX9 is strongly positively correlated with claudin 2. Finally, overexpression of SOX9 and claudin 2 will delay PM2.5-induced keratinocyte senescence. The silencing of claudin 2 leads to the loss of SOX9 function. It is clearly evident that SOX9 can affect the transcription of claudin 2, which increases first and then decreases in the process of mice from newborn to aging. SOX9 inhibits proinflammatory mediators, increases antioxidant capacity, and restores keratin differentiation. It can effectively prevent melanin deposition and delay aging.
Copyright © 2022 Jingyan Wang et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures
Similar articles
-
Expression and functional role of Sox9 in human epidermal keratinocytes.PLoS One. 2013;8(1):e54355. doi: 10.1371/journal.pone.0054355. Epub 2013 Jan 18. PLoS One. 2013. PMID: 23349860 Free PMC article.
-
microRNA-130a Promotes Human Keratinocyte Viability and Migration and Inhibits Apoptosis Through Direct Regulation of STK40-Mediated NF-κB Pathway and Indirect Regulation of SOX9-Meditated JNK/MAPK Pathway: A Potential Role in Psoriasis.DNA Cell Biol. 2017 Mar;36(3):219-226. doi: 10.1089/dna.2016.3517. Epub 2017 Jan 13. DNA Cell Biol. 2017. Retraction in: DNA Cell Biol. 2021 Jul;40(7):1037. doi: 10.1089/dna.2016.3517.retract. PMID: 28085489 Retracted.
-
Lhx2 differentially regulates Sox9, Tcf4 and Lgr5 in hair follicle stem cells to promote epidermal regeneration after injury.Development. 2011 Nov;138(22):4843-52. doi: 10.1242/dev.070284. Development. 2011. PMID: 22028024 Free PMC article.
-
The transcription factor Znf219 regulates chondrocyte differentiation by assembling a transcription factory with Sox9.J Cell Sci. 2010 Nov 1;123(Pt 21):3780-8. doi: 10.1242/jcs.071373. Epub 2010 Oct 12. J Cell Sci. 2010. PMID: 20940257
-
Diverse Regulation but Conserved Function: SOX9 in Vertebrate Sex Determination.Genes (Basel). 2021 Mar 26;12(4):486. doi: 10.3390/genes12040486. Genes (Basel). 2021. PMID: 33810596 Free PMC article. Review.
Cited by
-
A scATAC-seq atlas of stasis zone in rat skin burn injury wound process.Front Cell Dev Biol. 2025 Jan 7;12:1519926. doi: 10.3389/fcell.2024.1519926. eCollection 2024. Front Cell Dev Biol. 2025. PMID: 39845081 Free PMC article.
-
Marine Bioactive Peptides: Anti-Photoaging Mechanisms and Potential Skin Protective Effects.Curr Issues Mol Biol. 2024 Jan 23;46(2):990-1009. doi: 10.3390/cimb46020063. Curr Issues Mol Biol. 2024. PMID: 38392181 Free PMC article. Review.
References
-
- Cai X. W., Zhu R., Ran L., et al. A novel non-contact communication between human keratinocytes and T cells: Exosomes derived from keratinocytes support superantigen induced proliferation of resting T cells. Molecular Medicine Reports . 2017;16(5):7032–7038. doi: 10.3892/mmr.2017.7492. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
