Multicolor Nitrogen-Doped Carbon Quantum Dots for Environment-Dependent Emission Tuning
- PMID: 35967036
- PMCID: PMC9366982
- DOI: 10.1021/acsomega.2c03912
Multicolor Nitrogen-Doped Carbon Quantum Dots for Environment-Dependent Emission Tuning
Abstract
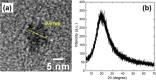
Carbon quantum dots (CQDs) have potential applications in many fields such as light-emitting devices, photocatalysis, and bioimaging due to their unique photoluminescence (PL) properties and environmental benignness. Here, we report the synthesis of nitrogen-doped carbon quantum dots (NCQDs) from citric acid and m-phenylenediamine using a one-pot hydrothermal approach. The environment-dependent emission changes of NCQDs were extensively investigated in various solvents, in the solid state, and in physically assembled PMMA-PnBA-PMMA copolymer gels in 2-ethyl-hexanol. NCQDs display bright emissions in various solvents as well as in the solid state. These NCQDs exhibit multicolor PL emission across the visible region upon changing the environment (solutions and polymer matrices). NCQDs also exhibit excitation-dependent PL and solvatochromism, which have not been frequently investigated in CQDs. Most CQDs are nonemissive in the aggregated or solid state due to the aggregation-caused quenching (ACQ) effect, limiting their solid-state applications. However, NCQDs synthesized here display a strong solid-state emission centered at 568 nm attributed to the presence of surface functional groups that restrict the π-π interaction between the NCQDs and assist in overcoming the ACQ effect in the solid state. NCQD-containing gels display significant fluorescence enhancement in comparison to the NCQDs in 2-ethyl hexanol, likely because of the interaction between the polar PMMA blocks and NCQDs. The application of NCQDs-Gel as a solid/gel state fluorescent display has been presented. This research facilitates the development of large-scale, low-cost multicolor phosphor for the fabrication of optoelectronic devices, sensing, and bioimaging applications.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures








Similar articles
-
Highly luminescent N-doped carbon quantum dots as an effective multifunctional fluorescence sensing platform.Chemistry. 2014 Feb 17;20(8):2254-63. doi: 10.1002/chem.201304374. Epub 2014 Jan 21. Chemistry. 2014. PMID: 24449509
-
Hydrothermal synthesis of nitrogen-doped carbon quantum dots from lignin for formaldehyde determination.RSC Adv. 2021 Sep 1;11(47):29178-29185. doi: 10.1039/d1ra05370a. eCollection 2021 Sep 1. RSC Adv. 2021. PMID: 35479568 Free PMC article.
-
Nitrogen-doped carbon quantum dots conjugated isoreticular metal-organic framework-3 particles based luminescent probe for selective sensing of trinitrotoluene explosive.Mikrochim Acta. 2020 Sep 1;187(9):536. doi: 10.1007/s00604-020-04496-0. Mikrochim Acta. 2020. PMID: 32870369
-
Carbon-Based Quantum Dots with Solid-State Photoluminescent: Mechanism, Implementation, and Application.Small. 2020 Dec;16(48):e2004621. doi: 10.1002/smll.202004621. Epub 2020 Nov 3. Small. 2020. PMID: 33145929 Review.
-
Green synthesis of carbon quantum dots and their environmental applications.Environ Res. 2022 Sep;212(Pt B):113283. doi: 10.1016/j.envres.2022.113283. Epub 2022 Apr 21. Environ Res. 2022. PMID: 35461844 Review.
Cited by
-
Water Soluble PMPC-Derived Bright Fluorescent Nitrogen/Phosphorous-Doped Carbon Dots for Fluorescent Ink (Anti-Counterfeiting) and Cellular Multicolor Imaging.Polymers (Basel). 2023 Mar 8;15(6):1352. doi: 10.3390/polym15061352. Polymers (Basel). 2023. PMID: 36987134 Free PMC article.
-
Manifestation of Donor-Acceptor Properties of N-Doped Polymer Carbon Dots During Hydrogen Bonds Formation in Different Solvents.Polymers (Basel). 2024 Dec 21;16(24):3585. doi: 10.3390/polym16243585. Polymers (Basel). 2024. PMID: 39771437 Free PMC article.
References
-
- Murray C. B.; Norris D. J.; Bawendi M. G. Synthesis and Characterization of Nearly Monodisperse CdE (E = Sulfur, Selenium, Tellurium) Semiconductor Nanocrystallites. J. Am. Chem. Soc. 1993, 115, 8706–8715. 10.1021/ja00072a025. - DOI
-
- Colvin V. L.; Schlamp M. C.; Alivisatos A. P. Light-Emitting Diodes Made from Cadmium Selenide Nanocrystals and a Semiconducting Polymer. Nature 1994, 370, 354–357. 10.1038/370354a0. - DOI
-
- Zhu S.; Song Y.; Zhao X.; Shao J.; Zhang J.; Yang B. The Photoluminescence Mechanism in Carbon Dots (Graphene Quantum Dots, Carbon Nanodots, and Polymer Dots): Current State and Future Perspective. Nano Res. 2015, 8, 355–381. 10.1007/s12274-014-0644-3. - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

