A temporo-spatial pharmacometabolomics method to characterize pharmacokinetics and pharmacodynamics in the brain microregions by using ambient mass spectrometry imaging
- PMID: 35967273
- PMCID: PMC9366215
- DOI: 10.1016/j.apsb.2022.03.018
A temporo-spatial pharmacometabolomics method to characterize pharmacokinetics and pharmacodynamics in the brain microregions by using ambient mass spectrometry imaging
Abstract
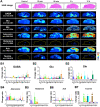
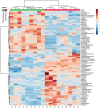
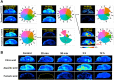
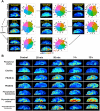
The brain is the most advanced organ with various complex structural and functional microregions. It is often challenging to understand what and where the molecular events would occur for a given drug treatment in the brain. Herein, a temporo-spatial pharmacometabolomics method was proposed based on ambient mass spectrometry imaging and was applied to evaluate the microregional effect of olanzapine (OLZ) on brain tissue and demonstrate its effectiveness in characterizing the microregional pharmacokinetics and pharmacodynamics of OLZ for improved understanding of the molecular mechanism of drugs acting on the microregions of the brain. It accurately and simultaneously illustrated the levels dynamics and microregional distribution of various substances, including exogenous drugs and its metabolites, as well as endogenous functional metabolites from complicated brain tissue. The targeted imaging analysis of the prototype drug and its metabolites presented the absorption, distribution, metabolism, and excretion characteristics of the drug itself. Moreover, the endogenous functional metabolites were identified along with the associated therapeutic and adverse effects of the drug, which can reflect the pharmacodynamics effect on the microregional brain. Therefore, this method is significant in elucidating and understanding the molecular mechanism of central nervous system drugs at the temporo and spatial metabolic level of system biology.
Keywords: Antipsychotic drug; Mass spectrometry imaging; Pharmacodynamics; Pharmacokinetics; Pharmacometabolomics.
© 2022 Chinese Pharmaceutical Association and Institute of Materia Medica, Chinese Academy of Medical Sciences. Production and hosting by Elsevier B.V.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
A targeted neurotransmitter quantification and nontargeted metabolic profiling method for pharmacometabolomics analysis of olanzapine by using UPLC-HRMS.RSC Adv. 2020 May 13;10(31):18305-18314. doi: 10.1039/d0ra02406f. eCollection 2020 May 10. RSC Adv. 2020. PMID: 35517196 Free PMC article.
-
Mapping Metabolic Networks in the Brain by Ambient Mass Spectrometry Imaging and Metabolomics.Anal Chem. 2021 May 4;93(17):6746-6754. doi: 10.1021/acs.analchem.1c00467. Epub 2021 Apr 23. Anal Chem. 2021. PMID: 33890766
-
Pharmacokinetics in Pharmacometabolomics: Towards Personalized Medication.Pharmaceuticals (Basel). 2023 Nov 7;16(11):1568. doi: 10.3390/ph16111568. Pharmaceuticals (Basel). 2023. PMID: 38004434 Free PMC article. Review.
-
Spatiotemporal pharmacometabolomics based on ambient mass spectrometry imaging to evaluate the metabolism and hepatotoxicity of amiodarone in HepG2 spheroids.J Pharm Anal. 2023 May;13(5):483-493. doi: 10.1016/j.jpha.2023.04.007. Epub 2023 Apr 14. J Pharm Anal. 2023. PMID: 37305784 Free PMC article.
-
Recent Progress on Mass Spectrum Based Approaches for Absorption, Distribution, Metabolism, and Excretion Characterization of Traditional Chinese Medicine.Curr Drug Metab. 2022;23(2):99-112. doi: 10.2174/1389200223666220211093548. Curr Drug Metab. 2022. PMID: 35152864 Review.
Cited by
-
Gut microbiota depletion and FXR inhibition exacerbates zonal hepatotoxicity of sunitinib.Theranostics. 2024 Oct 28;14(18):7219-7240. doi: 10.7150/thno.99926. eCollection 2024. Theranostics. 2024. PMID: 39629129 Free PMC article.
-
Localization analysis of metabolites from complex biological samples-recent analytical technique of mass spectrometry imaging.Front Mol Biosci. 2023 Apr 21;10:1169449. doi: 10.3389/fmolb.2023.1169449. eCollection 2023. Front Mol Biosci. 2023. PMID: 37152901 Free PMC article. No abstract available.
-
Spatial metabolomics highlights metabolic reprogramming in acute myeloid leukemia mice through creatine pathway.Acta Pharm Sin B. 2024 Oct;14(10):4461-4477. doi: 10.1016/j.apsb.2024.07.004. Epub 2024 Jul 6. Acta Pharm Sin B. 2024. PMID: 39525575 Free PMC article.
-
Druggability Studies of Benzene Sulfonamide Substituted Diarylamide (E3) as a Novel Diuretic.Biomedicines. 2025 Apr 18;13(4):992. doi: 10.3390/biomedicines13040992. Biomedicines. 2025. PMID: 40299675 Free PMC article.
-
Discovering metabolic vulnerability using spatially resolved metabolomics for antitumor small molecule-drug conjugates development as a precise cancer therapy strategy.J Pharm Anal. 2023 Jul;13(7):776-787. doi: 10.1016/j.jpha.2023.02.010. Epub 2023 Feb 28. J Pharm Anal. 2023. PMID: 37577390 Free PMC article.
References
-
- Park H.J., Friston K. Structural and functional brain networks: from connections to cognition. Science. 2013;342:1238411. - PubMed
-
- Zhao R., Pollack G.M. Regional differences in capillary density, perfusion rate, and P-glycoprotein activity: a quantitative analysis of regional drug exposure in the brain. Biochem Pharmacol. 2009;78:1052–1059. - PubMed
-
- Pang X., Gao S., Ga M., Zhang J., Luo Z., Chen Y., et al. Mapping metabolic networks in the brain by ambient mass spectrometry imaging and metabolomics. Anal Chem. 2021;93:6746–6754. - PubMed
LinkOut - more resources
Full Text Sources

