Biofilm formation and antibiotic sensitivity in Elizabethkingia anophelis
- PMID: 35967866
- PMCID: PMC9366890
- DOI: 10.3389/fcimb.2022.953780
Biofilm formation and antibiotic sensitivity in Elizabethkingia anophelis
Abstract
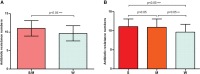
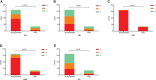
Elizabethkingia anophelis has recently gained global attention and is emerging as a cause of life-threatening nosocomial infections. The present study aimed to investigate the association between antimicrobial resistance and the ability to form biofilm among E. anophelis isolated from hospitalized patients in China. Over 10 years, a total of 197 non-duplicate E. anophelis strains were collected. Antibiotic susceptibility was determined by the standard agar dilution method as a reference assay according to the Clinical and Laboratory Standards Institute. The biofilm formation ability was assessed using a culture microtiter plate method, which was determined using a crystal violet assay. Culture plate results were cross-checked by scanning electron microscopy imaging analysis. Among the 197 isolates, all were multidrug-resistant, and 20 were extensively drug-resistant. Clinical E. anophelis showed high resistance to current antibiotics, and 99% of the isolates were resistant to at least seven antibiotics. The resistance rate for aztreonam, ceftazidime, imipenem, meropenem, trimethoprim-sulfamethoxazole, cefepime, and tetracycline was high as 100%, 99%, 99%, 99%, 99%, 95%, and 90%, respectively. However, the isolates exhibited the highest susceptibility to minocycline (100%), doxycycline (96%), and rifampin (94%). The biofilm formation results revealed that all strains could form biofilm. Among them, the proportions of strong, medium, and weak biofilm-forming strains were 41%, 42%, and 17%, respectively. Furthermore, the strains forming strong or moderate biofilm presented a statistically significant higher resistance than the weak formers (p < 0.05), especially for piperacillin, piperacillin-tazobactam, cefepime, amikacin, and ciprofloxacin. Although E. anophelis was notoriously resistant to large antibiotics, minocycline, doxycycline, and rifampin showed potent activity against this pathogen. The data in the present report revealed a positive association between biofilm formation and antibiotic resistance, which will provide a foundation for improved therapeutic strategies against E. anophelis infections in the future.
Keywords: Elizabethkingia anophelis; biofilm formation; biofilm-specific resistance; multidrug-resistant; nosocomial infections.
Copyright © 2022 Hu, Lv, Xu, Zheng and Xiao.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Antimicrobial Susceptibility Patterns and Antimicrobial Therapy of Infections Caused by Elizabethkingia Species.Medicina (Kaunas). 2024 Dec 2;60(12):1990. doi: 10.3390/medicina60121990. Medicina (Kaunas). 2024. PMID: 39768869 Free PMC article. Review.
-
Molecular characteristics and in vitro effects of antimicrobial combinations on planktonic and biofilm forms of Elizabethkingia anophelis.J Antimicrob Chemother. 2021 Apr 13;76(5):1205-1214. doi: 10.1093/jac/dkab018. J Antimicrob Chemother. 2021. PMID: 33532826
-
Susceptibility of Elizabethkingia spp. to commonly tested and novel antibiotics and concordance between broth microdilution and automated testing methods.J Antimicrob Chemother. 2021 Feb 11;76(3):653-658. doi: 10.1093/jac/dkaa499. J Antimicrob Chemother. 2021. PMID: 33258923 Free PMC article.
-
Molecular identification and biofilm-forming ability of Elizabethkingia species.Microb Pathog. 2022 Jan;162:105345. doi: 10.1016/j.micpath.2021.105345. Epub 2021 Dec 9. Microb Pathog. 2022. PMID: 34896547
-
Emerging infections in vulnerable hosts: Stenotrophomonas maltophilia and Elizabethkingia anophelis.Curr Opin Infect Dis. 2023 Dec 1;36(6):481-494. doi: 10.1097/QCO.0000000000000953. Epub 2023 Aug 2. Curr Opin Infect Dis. 2023. PMID: 37548375
Cited by
-
Microbiological Characterization and Clinical Facets of Elizabethkingia Bloodstream Infections in a Tertiary Care Hospital of Eastern India.Infect Drug Resist. 2023 May 24;16:3257-3267. doi: 10.2147/IDR.S409121. eCollection 2023. Infect Drug Resist. 2023. PMID: 37249959 Free PMC article.
-
Antibiotic Resistance and Virulence Factors in Clinical Isolates of Stenotrophomonas maltophilia from Hospitalized Patients in Tehran, Iran.Int J Microbiol. 2024 Oct 1;2024:8224242. doi: 10.1155/2024/8224242. eCollection 2024. Int J Microbiol. 2024. PMID: 39380784 Free PMC article.
-
Antimicrobial Susceptibility Patterns and Antimicrobial Therapy of Infections Caused by Elizabethkingia Species.Medicina (Kaunas). 2024 Dec 2;60(12):1990. doi: 10.3390/medicina60121990. Medicina (Kaunas). 2024. PMID: 39768869 Free PMC article. Review.
-
Central Line-Associated Bloodstream Infection Due to Elizabethkingia anophelis: Case Report and Literature Review on Pediatric Infections.Microorganisms. 2024 Jun 4;12(6):1145. doi: 10.3390/microorganisms12061145. Microorganisms. 2024. PMID: 38930527 Free PMC article.
-
Structural and functional characterization of cyclic pyrimidine-regulated anti-phage system.Nat Commun. 2024 Jul 4;15(1):5634. doi: 10.1038/s41467-024-49861-2. Nat Commun. 2024. PMID: 38965224 Free PMC article.
References
-
- Burnard D., Gore L., Henderson A., Ranasinghe A., Bergh H., Cottrell K., et al. . (2020). Comparative genomics and antimicrobial resistance profiling of Elizabethkingia isolates reveal nosocomial transmission and In vitro susceptibility to fluoroquinolones, tetracyclines, and trimethoprim-sulfamethoxazole. J. Clin. Microbiol. 58, e00730–20. doi: 10.1128/JCM.00730-20 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical

