EEG biomarkers for the diagnosis and treatment of infantile spasms
- PMID: 35968272
- PMCID: PMC9366674
- DOI: 10.3389/fneur.2022.960454
EEG biomarkers for the diagnosis and treatment of infantile spasms
Abstract
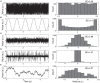
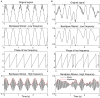
Early diagnosis and treatment are critical for young children with infantile spasms (IS), as this maximizes the possibility of the best possible child-specific outcome. However, there are major barriers to achieving this, including high rates of misdiagnosis or failure to recognize the seizures, medication failure, and relapse. There are currently no validated tools to aid clinicians in assessing objective diagnostic criteria, predicting or measuring medication response, or predicting the likelihood of relapse. However, the pivotal role of EEG in the clinical management of IS has prompted many recent studies of potential EEG biomarkers of the disease. These include both visual EEG biomarkers based on human visual interpretation of the EEG and computational EEG biomarkers in which computers calculate quantitative features of the EEG. Here, we review the literature on both types of biomarkers, organized based on the application (diagnosis, treatment response, prediction, etc.). Visual biomarkers include the assessment of hypsarrhythmia, epileptiform discharges, fast oscillations, and the Burden of AmplitudeS and Epileptiform Discharges (BASED) score. Computational markers include EEG amplitude and power spectrum, entropy, functional connectivity, high frequency oscillations (HFOs), long-range temporal correlations, and phase-amplitude coupling. We also introduce each of the computational measures and provide representative examples. Finally, we highlight remaining gaps in the literature, describe practical guidelines for future biomarker discovery and validation studies, and discuss remaining roadblocks to clinical implementation, with the goal of facilitating future work in this critical area.
Keywords: Detrended Fluctuation Analysis (DFA); entropy; epilepsy; functional connectivity; high frequency oscillation (HFO); hypsarrhythmia; long-range temporal correlation (LRTC); phase amplitude coupling.
Copyright © 2022 Romero Milà, Remakanthakurup Sindhu, Mytinger, Shrey and Lopour.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
-
- Specchio N, Wirrell EC, Scheffer IE, Nabbout R, Riney K, Samia P, et al. . International league against epilepsy classification and definition of epilepsy syndromes with onset in childhood: position paper by the ILAE task force on nosology and definitions. Epilepsia. (2022) 5:1–49. 10.1111/epi.17241 - DOI - PubMed

