The Notch signaling pathway in skeletal muscle health and disease
- PMID: 35968817
- PMCID: PMC9804383
- DOI: 10.1002/mus.27684
The Notch signaling pathway in skeletal muscle health and disease
Abstract
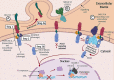
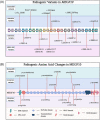
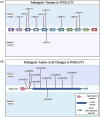
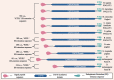
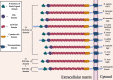
The Notch signaling pathway is a key regulator of skeletal muscle development and regeneration. Over the past decade, the discoveries of three new muscle disease genes have added a new dimension to the relationship between the Notch signaling pathway and skeletal muscle: MEGF10, POGLUT1, and JAG2. We review the clinical syndromes associated with pathogenic variants in each of these genes, known molecular and cellular functions of their protein products with a particular focus on the Notch signaling pathway, and potential novel therapeutic targets that may emerge from further investigations of these diseases. The phenotypes associated with two of these genes, POGLUT1 and JAG2, clearly fall within the realm of muscular dystrophy, whereas the third, MEGF10, is associated with a congenital myopathy/muscular dystrophy overlap syndrome classically known as early-onset myopathy, areflexia, respiratory distress, and dysphagia. JAG2 is a canonical Notch ligand, POGLUT1 glycosylates the extracellular domain of Notch receptors, and MEGF10 interacts with the intracellular domain of NOTCH1. Additional genes and their encoded proteins relevant to muscle function and disease with links to the Notch signaling pathway include TRIM32, ATP2A1 (SERCA1), JAG1, PAX7, and NOTCH2NLC. There is enormous potential to identify convergent mechanisms of skeletal muscle disease and new therapeutic targets through further investigations of the Notch signaling pathway in the context of skeletal muscle development, maintenance, and disease.
Keywords: JAG2; MEGF10; Notch signaling pathway; POGLUT1; muscular dystrophy.
© 2022 The Authors. Muscle & Nerve published by Wiley Periodicals LLC.
Conflict of interest statement
None of the authors has any conflict of interest to disclose.
Figures

Similar articles
-
A form of muscular dystrophy associated with pathogenic variants in JAG2.Am J Hum Genet. 2021 May 6;108(5):840-856. doi: 10.1016/j.ajhg.2021.03.020. Epub 2021 Apr 15. Am J Hum Genet. 2021. PMID: 33861953 Free PMC article.
-
A POGLUT1 mutation causes a muscular dystrophy with reduced Notch signaling and satellite cell loss.EMBO Mol Med. 2016 Nov 2;8(11):1289-1309. doi: 10.15252/emmm.201505815. Print 2016 Nov. EMBO Mol Med. 2016. PMID: 27807076 Free PMC article.
-
POGLUT1 biallelic mutations cause myopathy with reduced satellite cells, α-dystroglycan hypoglycosylation and a distinctive radiological pattern.Acta Neuropathol. 2020 Mar;139(3):565-582. doi: 10.1007/s00401-019-02117-6. Epub 2020 Jan 3. Acta Neuropathol. 2020. PMID: 31897643 Free PMC article.
-
Diseases related to Notch glycosylation.Mol Aspects Med. 2021 Jun;79:100938. doi: 10.1016/j.mam.2020.100938. Epub 2020 Dec 16. Mol Aspects Med. 2021. PMID: 33341260 Review.
-
Notch signaling pathway in osteogenesis, bone development, metabolism, and diseases.FASEB J. 2025 Feb 28;39(4):e70417. doi: 10.1096/fj.202402545R. FASEB J. 2025. PMID: 39985304 Review.
Cited by
-
Transcriptome Data Revealed the circRNA-miRNA-mRNA Regulatory Network during the Proliferation and Differentiation of Myoblasts in Shitou Goose.Animals (Basel). 2024 Feb 8;14(4):576. doi: 10.3390/ani14040576. Animals (Basel). 2024. PMID: 38396545 Free PMC article.
-
Congenital myopathies: pathophysiological mechanisms and promising therapies.J Transl Med. 2024 Sep 2;22(1):815. doi: 10.1186/s12967-024-05626-5. J Transl Med. 2024. PMID: 39223631 Free PMC article. Review.
-
Tissue-based T cell activation and viral RNA persist for up to 2 years after SARS-CoV-2 infection.Sci Transl Med. 2024 Jul 3;16(754):eadk3295. doi: 10.1126/scitranslmed.adk3295. Epub 2024 Jul 3. Sci Transl Med. 2024. PMID: 38959327 Free PMC article.
-
Case Report: Exploring the clinical spectrum of LGMD R27: insights from a case study with homozygous pathogenic variant in the JAG2 gene.Front Pediatr. 2024 Nov 22;12:1414465. doi: 10.3389/fped.2024.1414465. eCollection 2024. Front Pediatr. 2024. PMID: 39649397 Free PMC article.
-
Decoding the NRF2-NOTCH Crosstalk in Lung Cancer-An Update.Antioxidants (Basel). 2025 May 29;14(6):657. doi: 10.3390/antiox14060657. Antioxidants (Basel). 2025. PMID: 40563292 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

