Salmonella Typhimurium outer membrane protein A (OmpA) renders protection from nitrosative stress of macrophages by maintaining the stability of bacterial outer membrane
- PMID: 35969640
- PMCID: PMC9410544
- DOI: 10.1371/journal.ppat.1010708
Salmonella Typhimurium outer membrane protein A (OmpA) renders protection from nitrosative stress of macrophages by maintaining the stability of bacterial outer membrane
Abstract
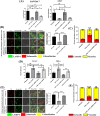
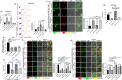
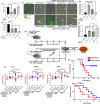
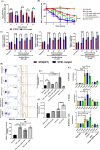
Bacterial porins are highly conserved outer membrane proteins used in the selective transport of charged molecules across the membrane. In addition to their significant contributions to the pathogenesis of Gram-negative bacteria, their role(s) in salmonellosis remains elusive. In this study, we investigated the role of outer membrane protein A (OmpA), one of the major outer membrane porins of Salmonella, in the pathogenesis of Salmonella Typhimurium (STM). Our study revealed that OmpA plays an important role in the intracellular virulence of Salmonella. An ompA deficient strain of Salmonella (STM ΔompA) showed compromised proliferation in macrophages. We found that the SPI-2 encoded virulence factors such as sifA and ssaV are downregulated in STM ΔompA. The poor colocalization of STM ΔompA with LAMP-1 showed that disruption of SCV facilitated its release into the cytosol of macrophages, where it was assaulted by reactive nitrogen intermediates (RNI). The enhanced recruitment of nitrotyrosine on the cytosolic population of STM ΔompAΔsifA and ΔompAΔssaV compared to STM ΔsifA and ΔssaV showed an additional role of OmpA in protecting the bacteria from host nitrosative stress. Further, we showed that the generation of greater redox burst could be responsible for enhanced sensitivity of STM ΔompA to the nitrosative stress. The expression of several other outer membrane porins such as ompC, ompD, and ompF was upregulated in STM ΔompA. We found that in the absence of ompA, the enhanced expression of ompF increased the outer membrane porosity of Salmonella and made it susceptible to in vitro and in vivo nitrosative stress. Our study illustrates a novel mechanism for the strategic utilization of OmpA by Salmonella to protect itself from the nitrosative stress of macrophages.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




Similar articles
-
Outer Membrane Porins Contribute to Antimicrobial Resistance in Gram-Negative Bacteria.Microorganisms. 2023 Jun 28;11(7):1690. doi: 10.3390/microorganisms11071690. Microorganisms. 2023. PMID: 37512863 Free PMC article. Review.
-
Functional OmpA of Salmonella Typhimurium Provides Protection From Lysosomal Degradation and Inhibits Autophagic Processes in Macrophages.J Infect Dis. 2025 Mar 17;231(3):716-728. doi: 10.1093/infdis/jiae376. J Infect Dis. 2025. PMID: 39078938
-
Loss of outer membrane protein A (OmpA) impairs the survival of Salmonella Typhimurium by inducing membrane damage in the presence of ceftazidime and meropenem.J Antimicrob Chemother. 2022 Nov 28;77(12):3376-3389. doi: 10.1093/jac/dkac327. J Antimicrob Chemother. 2022. PMID: 36177811
-
Functional analysis of OmpA and its contribution to pathogenesis of Edwardsiella tarda.Microb Pathog. 2024 Aug;193:106760. doi: 10.1016/j.micpath.2024.106760. Epub 2024 Jun 22. Microb Pathog. 2024. PMID: 38914348
-
Gram-Negative Bacterial Envelope Homeostasis under Oxidative and Nitrosative Stress.Microorganisms. 2022 Apr 28;10(5):924. doi: 10.3390/microorganisms10050924. Microorganisms. 2022. PMID: 35630368 Free PMC article. Review.
Cited by
-
Immunoinformatics design of multi-epitope vaccine using OmpA, OmpD and enterotoxin against non-typhoidal salmonellosis.BMC Bioinformatics. 2023 Feb 24;24(1):63. doi: 10.1186/s12859-023-05183-6. BMC Bioinformatics. 2023. PMID: 36823524 Free PMC article.
-
TLR-mediated aggresome-like induced structures comprise antimicrobial peptides and attenuate intracellular bacterial survival.Mol Biol Cell. 2024 Mar 1;35(3):ar34. doi: 10.1091/mbc.E23-09-0347. Epub 2024 Jan 3. Mol Biol Cell. 2024. PMID: 38170582 Free PMC article.
-
Outer Membrane Porins Contribute to Antimicrobial Resistance in Gram-Negative Bacteria.Microorganisms. 2023 Jun 28;11(7):1690. doi: 10.3390/microorganisms11071690. Microorganisms. 2023. PMID: 37512863 Free PMC article. Review.
-
Genomic insights of Salmonella isolated from dry fermented sausage production chains in Spain and France.Sci Rep. 2024 May 22;14(1):11660. doi: 10.1038/s41598-024-62141-9. Sci Rep. 2024. PMID: 38777847 Free PMC article.
-
Reverse vaccinology-based identification and in silico characterization of immunogenic membrane proteins of Salmonella Typhimurium as novel vaccine targets against multidrug-resistant infections.BMC Microbiol. 2025 Aug 23;25(1):542. doi: 10.1186/s12866-025-04124-y. BMC Microbiol. 2025. PMID: 40849649 Free PMC article.
References
-
- March C, Moranta D, Regueiro V, Llobet E, Tomas A, Garmendia J, et al.. Klebsiella pneumoniae outer membrane protein A is required to prevent the activation of airway epithelial cells. J Biol Chem. 2011;286(12):9956–67. doi: 10.1074/jbc.M110.181008 ; PubMed Central PMCID: PMC3060550. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

