A sequential two-step priming scheme reproduces diversity in synaptic strength and short-term plasticity
- PMID: 35969787
- PMCID: PMC9407230
- DOI: 10.1073/pnas.2207987119
A sequential two-step priming scheme reproduces diversity in synaptic strength and short-term plasticity
Abstract
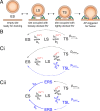
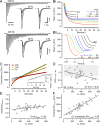
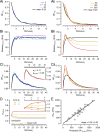
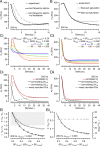
Glutamatergic synapses display variable strength and diverse short-term plasticity (STP), even for a given type of connection. Using nonnegative tensor factorization and conventional state modeling, we demonstrate that a kinetic scheme consisting of two sequential and reversible steps of release-machinery assembly and a final step of synaptic vesicle (SV) fusion reproduces STP and its diversity among synapses. Analyzing transmission at the calyx of Held synapses reveals that differences in synaptic strength and STP are not primarily caused by variable fusion probability (pfusion) but are determined by the fraction of docked synaptic vesicles equipped with a mature release machinery. Our simulations show that traditional quantal analysis methods do not necessarily report pfusion of SVs with a mature release machinery but reflect both pfusion and the distribution between mature and immature priming states at rest. Thus, the approach holds promise for a better mechanistic dissection of the roles of presynaptic proteins in the sequence of SV docking, two-step priming, and fusion. It suggests a mechanism for activity-induced redistribution of synaptic efficacy.
Keywords: calyx of Held; numerical simulation; short-term plasticity; synaptic transmission; synaptic vesicle priming.
Conflict of interest statement
The authors declare no competing interest.
Figures

Similar articles
-
Different states of synaptic vesicle priming explain target cell type-dependent differences in neurotransmitter release.Proc Natl Acad Sci U S A. 2024 Apr 30;121(18):e2322550121. doi: 10.1073/pnas.2322550121. Epub 2024 Apr 24. Proc Natl Acad Sci U S A. 2024. PMID: 38657053 Free PMC article.
-
Number and relative abundance of synaptic vesicles in functionally distinct priming states determine synaptic strength and short-term plasticity.J Physiol. 2025 Mar 22. doi: 10.1113/JP286282. Online ahead of print. J Physiol. 2025. PMID: 40120134
-
Regulation of a subset of release-ready vesicles by the presynaptic protein Mover.Proc Natl Acad Sci U S A. 2021 Jan 19;118(3):e2022551118. doi: 10.1073/pnas.2022551118. Proc Natl Acad Sci U S A. 2021. PMID: 33431696 Free PMC article.
-
Dynamically Primed Synaptic Vesicle States: Key to Understand Synaptic Short-Term Plasticity.Neuron. 2018 Dec 19;100(6):1283-1291. doi: 10.1016/j.neuron.2018.11.024. Neuron. 2018. PMID: 30571941 Review.
-
Regulation of synaptic release-site Ca2+ channel coupling as a mechanism to control release probability and short-term plasticity.FEBS Lett. 2018 Nov;592(21):3516-3531. doi: 10.1002/1873-3468.13188. Epub 2018 Jul 31. FEBS Lett. 2018. PMID: 29993122 Review.
Cited by
-
Different states of synaptic vesicle priming explain target cell type-dependent differences in neurotransmitter release.Proc Natl Acad Sci U S A. 2024 Apr 30;121(18):e2322550121. doi: 10.1073/pnas.2322550121. Epub 2024 Apr 24. Proc Natl Acad Sci U S A. 2024. PMID: 38657053 Free PMC article.
-
Pre- versus Post-synaptic Forms of LTP in Two Branches of the Same Hippocampal Afferent.J Neurosci. 2024 Mar 6;44(10):e1449232024. doi: 10.1523/JNEUROSCI.1449-23.2024. J Neurosci. 2024. PMID: 38326038 Free PMC article.
-
Estimates of quantal synaptic parameters in light of more complex vesicle pool models.Front Cell Neurosci. 2025 Mar 18;19:1556360. doi: 10.3389/fncel.2025.1556360. eCollection 2025. Front Cell Neurosci. 2025. PMID: 40170993 Free PMC article.
-
Cav2.2 Channels Sustain Vesicle Recruitment at a Mature Glutamatergic Synapse.J Neurosci. 2023 May 31;43(22):4005-4018. doi: 10.1523/JNEUROSCI.1279-22.2023. Epub 2023 Apr 26. J Neurosci. 2023. PMID: 37185239 Free PMC article.
-
Parallel processing of quickly and slowly mobilized reserve vesicles in hippocampal synapses.Elife. 2024 May 10;12:RP88212. doi: 10.7554/eLife.88212. Elife. 2024. PMID: 38727712 Free PMC article.
References
-
- Abbott L. F., Regehr W. G., Synaptic computation. Nature 431, 796–803 (2004). - PubMed
-
- Bartos M., Vida I., Jonas P., Synaptic mechanisms of synchronized gamma oscillations in inhibitory interneuron networks. Nat. Rev. Neurosci. 8, 45–56 (2007). - PubMed
-
- Fortune E. S., Rose G. J., Short-term synaptic plasticity as a temporal filter. Trends Neurosci. 24, 381–385 (2001). - PubMed
-
- Verhage M., Sørensen J. B., SNAREopathies: Diversity in mechanisms and symptoms. Neuron 107, 22–37 (2020). - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

