A common SNP in Chrna5 enhances morphine reward in female mice
- PMID: 35973602
- PMCID: PMC12120996
- DOI: 10.1016/j.neuropharm.2022.109218
A common SNP in Chrna5 enhances morphine reward in female mice
Abstract
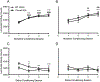
The single nucleotide polymorphism (SNP) D398N (rs16969968) in CHRNA5, the gene encoding the α5 subunit of the nicotinic acetylcholine receptors (nAChR), has been associated with both nicotine and opiate dependence in human populations. Expression of this SNP on presynaptic VTA dopaminergic (DA) neurons is known to cause a reduction in calcium signaling, leading to alterations in transmitter signaling and altered responses to drugs of abuse. To examine the impact of the Chrna5 SNP on opiate reward and underlying dopaminergic mechanisms, mice harboring two copies of the risk-associated allele (Chrna5 A/A) at a location equivalent to human rs16969968 were generated via CRISPR/cas9 genome editing. We sought to determine whether Chrna5 A/A mice show differences in sensitivity to rewarding properties of morphine using the conditioned place preference paradigm. When mice were tested two weeks after conditioning, female Chrna5 A/A mice showed significantly enhanced preference for the morphine-paired chamber relative to WT females, suggesting that this genotype may enhance opioid reward specifically in females. In contrast, Chrna5 genotype had no effect on locomotor sensitization in male or female mice. Relative to WT females, peak amplitude of ACh-gated currents recorded from VTA DA neurons in Chrna5 A/A females was potentiated 1 day after conditioning with morphine. Increased FOS expression was also observed in Chrna5 A/A mice relative to WT mice following exposure to the morphine CPP chamber. We propose that impaired α5 nAChR subunit function alters DA neuron response following repeated morphine exposures, and that this early cellular response could contribute to enhanced opiate reward two weeks after conditioning.
Keywords: Conditioned place preference; Mice; Morphine; Nicotinic acetylcholine receptor; Reward; Single nucleotide polymorphism.
Copyright © 2022 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of competing interests
None.
Figures

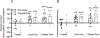


Similar articles
-
CHRNA5 gene variation affects the response of VTA dopaminergic neurons during chronic nicotine exposure and withdrawal.Neuropharmacology. 2023 Sep 1;235:109547. doi: 10.1016/j.neuropharm.2023.109547. Epub 2023 Apr 27. Neuropharmacology. 2023. PMID: 37116611 Free PMC article.
-
β4-Nicotinic Receptors Are Critically Involved in Reward-Related Behaviors and Self-Regulation of Nicotine Reinforcement.J Neurosci. 2020 Apr 22;40(17):3465-3477. doi: 10.1523/JNEUROSCI.0356-19.2020. Epub 2020 Mar 17. J Neurosci. 2020. PMID: 32184221 Free PMC article.
-
Mutation of the α5 nicotinic acetylcholine receptor subunit increases ethanol and nicotine consumption in adolescence and impacts adult drug consumption.Neuropharmacology. 2022 Sep 15;216:109170. doi: 10.1016/j.neuropharm.2022.109170. Epub 2022 Jun 22. Neuropharmacology. 2022. PMID: 35752273 Free PMC article.
-
Contribution of Variants in CHRNA5/A3/B4 Gene Cluster on Chromosome 15 to Tobacco Smoking: From Genetic Association to Mechanism.Mol Neurobiol. 2016 Jan;53(1):472-484. doi: 10.1007/s12035-014-8997-x. Epub 2014 Dec 5. Mol Neurobiol. 2016. PMID: 25471942 Review.
-
Linking the CHRNA5 SNP to drug abuse liability: From circuitry to cellular mechanisms.Neuropharmacology. 2021 Mar 15;186:108480. doi: 10.1016/j.neuropharm.2021.108480. Epub 2021 Feb 1. Neuropharmacology. 2021. PMID: 33539855 Free PMC article. Review.
Cited by
-
Opioid trail: Tracking contributions to opioid use disorder from host genetics to the gut microbiome.Neurosci Biobehav Rev. 2024 Jan;156:105487. doi: 10.1016/j.neubiorev.2023.105487. Epub 2023 Nov 30. Neurosci Biobehav Rev. 2024. PMID: 38040073 Free PMC article. Review.
-
Establishment and Application of a Novel Genetic Detection Panel for SNPs in Mongolian Gerbils.Genes (Basel). 2024 Jun 20;15(6):817. doi: 10.3390/genes15060817. Genes (Basel). 2024. PMID: 38927752 Free PMC article.
-
Development of a single-nucleotide polymorphism panel genotyping system for genetic analysis of Chinese hamsters.Animal Model Exp Med. 2025 May;8(5):916-921. doi: 10.1002/ame2.70006. Epub 2025 Feb 27. Animal Model Exp Med. 2025. PMID: 40016881 Free PMC article.
-
Distinct Chrna5 mutations link excessive alcohol use to types I/II vulnerability profiles and IPN GABAergic neurons.Transl Psychiatry. 2024 Nov 6;14(1):461. doi: 10.1038/s41398-024-03164-8. Transl Psychiatry. 2024. PMID: 39505853 Free PMC article.
References
-
- Azam L, Winzer-Serhan UH, Chen Y, Leslie FM, 2002. Expression of neuronal nicotinic acetylcholine receptor subunit mRNAs within midbrain dopamine neurons. J. Comp. Neurol. 444, 260–274. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

