A non-metabolic function of hexokinase 2 in small cell lung cancer: promotes cancer cell stemness by increasing USP11-mediated CD133 stability
- PMID: 35975322
- PMCID: PMC9558687
- DOI: 10.1002/cac2.12351
A non-metabolic function of hexokinase 2 in small cell lung cancer: promotes cancer cell stemness by increasing USP11-mediated CD133 stability
Abstract
Background: Maintenance of cancer stem-like cell (CSC) stemness supported by aberrantly regulated cancer cell metabolism is critical for CSC self-renewal and tumor progression. As a key glycolytic enzyme, hexokinase 2 (HK2) plays an instrumental role in aerobic glycolysis and tumor progression. However, whether HK2 directly contribute to CSC stemness maintenance in small cell lung cancer (SCLC) is largely unclear. In this study, we aimed to investgate whether HK2 independent of its glycolytic activity is directly involved in stemness maintenance of CSC in SCLC.
Methods: Immunoblotting analyses were conducted to determine the expression of HK2 in SCLC CSCs and their differentiated counterparts. CSC-like properties and tumorigenesis of SCLC cells with or without HK2 depletion or overexpression were examined by sphere formation assay and xenograft mouse model. Immunoprecipitation and mass spectrometry analyses were performed to identify the binding proteins of CD133. The expression levels of CD133-associated and CSC-relevant proteins were evaluated by immunoblotting, immunoprecipitation, immunofluorescence, and immunohistochemistry assay. RNA expression levels of Nanog, POU5F1, Lin28, HK2, Prominin-1 were analyzed through quantitative reverse transcription PCR. Polyubiquitination of CD133 was examined by in vitro or in vivo ubiquitination assay. CD133+ cells were sorted by flow cytometry using an anti-CD133 antibody.
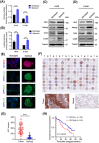
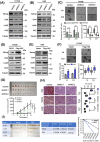
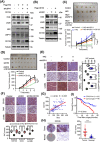
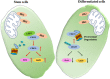
Results: We demonstrated that HK2 expression was much higher in CSCs of SCLC than in their differentiated counterparts. HK2 depletion inhibited CSC stemness and promoted CSC differentiation. Mechanistically, non-mitochondrial HK2 directly interacted with CD133 and enhanced CD133 expression without affecting CD133 mRNA levels. The interaction of HK2 and CD133 promoted the binding of the deubiquitinase ubiquitin-specific protease 11 (USP11) to CD133, thereby inhibiting CD133 polyubiquitylation and degradation. HK2-mediated upregulation of CD133 expression enhanced the expression of cell renewal regulators, SCLC cell stemness, and tumor growth in mice. In addition, HK2 expression was positively correlated with CD133 expression in human SCLC specimens, and their expression levels were associated with poor prognosis of SCLC patients.
Conclusions: These results revealed a critical non-metabolic function of HK2 in promotion of cancer cell stemness. Our findings provided new insights into the multifaceted roles of HK2 in tumor development.
Keywords: CD133; HK2; SCLC; USP11; cancer stem-like cell; metabolic enzyme; non-metabolic function; ubiquitylation.
© 2022 The Authors. Cancer Communications published by John Wiley & Sons Australia, Ltd. on behalf of Sun Yat-sen University Cancer Center.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures


References
-
- van Meerbeeck JP, Fennell DA, De Ruysscher DK. Small‐cell lung cancer. Lancet. 2011;378(9804):1741–55. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

