Brief Report: Virologic Impact of the Dolutegravir Transition: Prospective Results From the Multinational African Cohort Study
- PMID: 35980350
- PMCID: PMC9561234
- DOI: 10.1097/QAI.0000000000003065
Brief Report: Virologic Impact of the Dolutegravir Transition: Prospective Results From the Multinational African Cohort Study
Abstract
Background: The real-world impact on viral suppression of switching from non-dolutegravir-based therapy to tenofovir/lamivudine/dolutegravir (TLD) is not thoroughly characterized in Africa. We described the virologic consequences of switching regimens in the African Cohort Study (AFRICOS), an observational cohort in Nigeria, Kenya, Uganda, and Tanzania.
Methods: Among antiretroviral-experienced people living with HIV (PLWH) in AFRICOS, we compared viral load (VL) nonsuppression (VL ≥ 1000 copies/mL) among those who switched with those who never switched to TLD, restricting to participants who had at least 1 visit with a recorded VL after the countrywide rollout of TLD. We calculated Kaplan-Meier curves and conducted Cox proportional hazards modeling to estimate adjusted hazard ratios and 95% confidence intervals for factors potentially associated with nonsuppression.
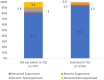
Results: As of September 1, 2021, there were 3108 PLWH enrolled. Among 1576 participants who switched to TLD, 1486 (94.3%) remained suppressed after transition, 12 (0.8%) remained unsuppressed, and 38 (2.4%) lost suppression, compared with 652 (82.1%), 75 (9.4%), and 46 (5.8%), respectively, of 797 participants who did not switch ( P < 0.001). After adjustment for sex, age, study site, and self-reported antiretroviral therapy adherence, virally suppressed participants who did not switch to TLD had significantly higher rates of losing viral suppression compared with those who switched (adjusted hazard ratio: 4.26; 95% confidence interval: 2.72 to 6.68).
Conclusions: PLWH transitioning to TLD had higher rates of viral suppression compared with those who remained on other regimens. Even within a highly suppressed population, TLD transition provided significant benefits for achieving or maintaining viral suppression.
Copyright © 2022 The Author(s). Published by Wolters Kluwer Health, Inc.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures
References
-
- Paton NI, Musaazi J, Sc M, et al. . Dolutegravir or darunavir in combination with zidovudine or tenofovir to treat HIV. N Engl J Med. 2021;385:330–341. - PubMed
-
- Aboud M, Kaplan R, Lombaard J, et al. . Dolutegravir versus ritonavir-boosted lopinavir both with dual nucleoside reverse transcriptase inhibitor therapy in adults with HIV-1 infection in whom first-line therapy has failed (DAWNING): an open-label, non-inferiority, phase 3b trial. Lancet Infect Dis. 2019;19:253–264. - PubMed
-
- Venter WDF, Moorhouse M, Sokhela S, et al. . Dolutegravir plus two different prodrugs of tenofovir to treat HIV. N Engl J Med. 2019;381:803–815. - PubMed
-
- Walmsley SL, Antela A, Clumeck N, et al. . Dolutegravir plus abacavir–lamivudine for the treatment of HIV-1 infection. N Engl J Med. 2013;369:1807–1818. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials