Topology and Excited State Multiplicity as Controlling Factors in the Carbazole-Photosensitized CPD Formation and Repair
- PMID: 35980822
- PMCID: PMC9447287
- DOI: 10.1021/acs.joc.2c00942
Topology and Excited State Multiplicity as Controlling Factors in the Carbazole-Photosensitized CPD Formation and Repair
Abstract
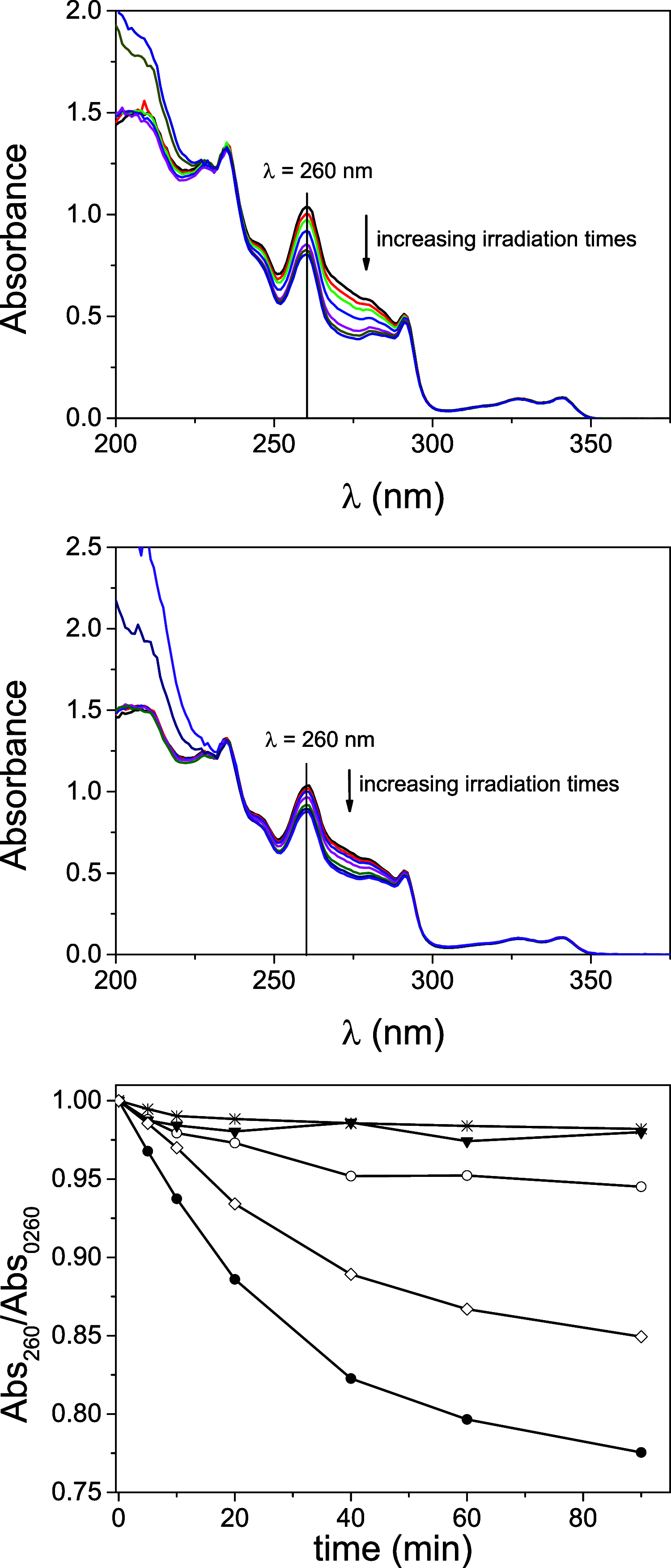
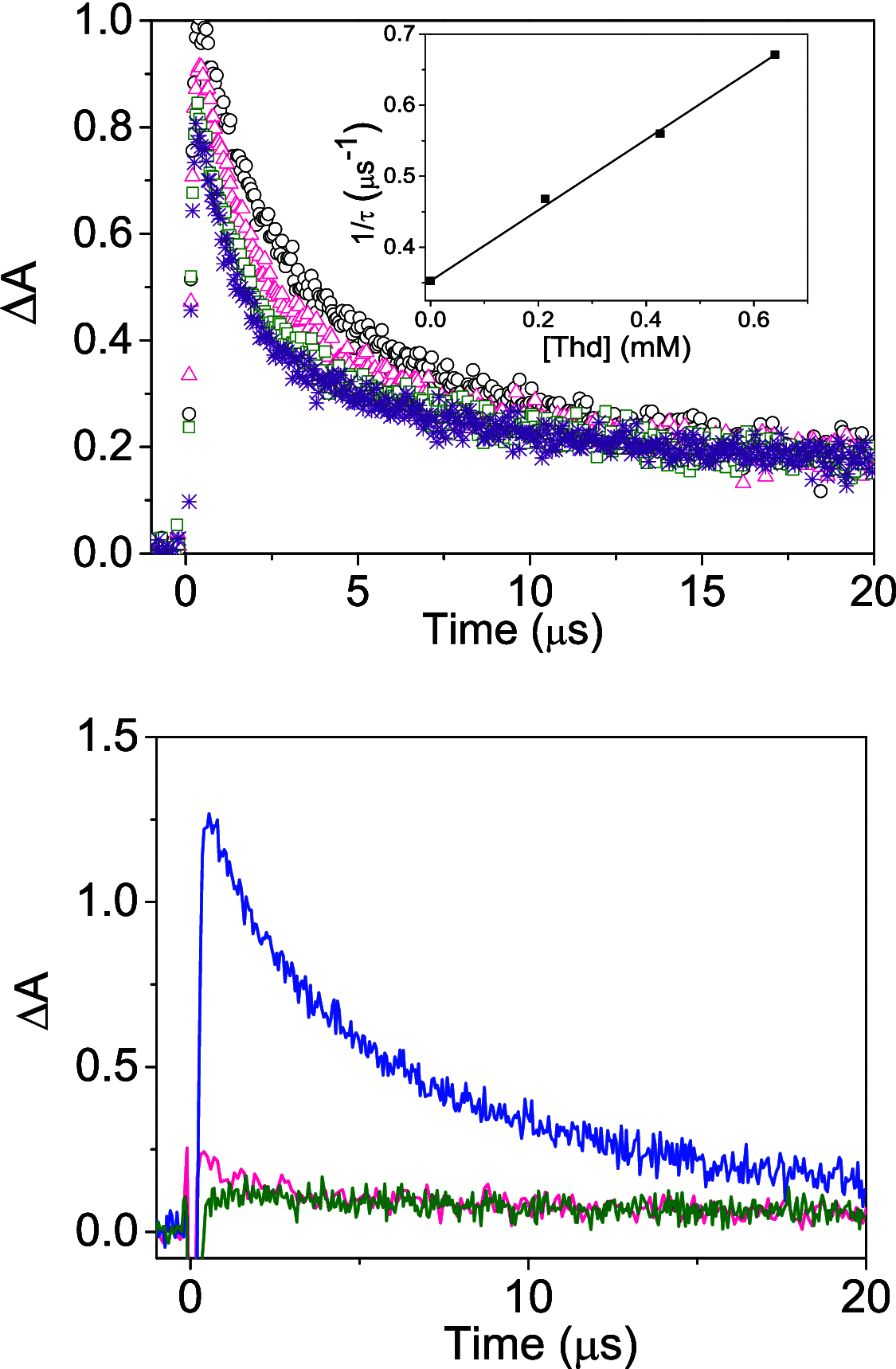
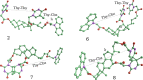
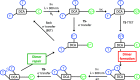
Photosensitized thymine<>thymine (Thy<>Thy) formation and repair can be mediated by carbazole (Cbz). The former occurs from the Cbz triplet excited state via energy transfer, while the latter takes place from the singlet excited state via electron transfer. Here, fundamental insight is provided into the role of the topology and excited state multiplicity, as factors governing the balance between both processes. This has been achieved upon designing and synthesizing different isomers of trifunctional systems containing one Cbz and two Thy units covalently linked to the rigid skeleton of the natural deoxycholic acid. The results shown here prove that the Cbz photosensitized dimerization is not counterbalanced by repair when the latter, instead of operating through-space, has to proceed through-bond.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Photosensitized Thymine Dimerization via Delocalized Triplet Excited States.Chemistry. 2015 Nov 16;21(47):17051-6. doi: 10.1002/chem.201502719. Epub 2015 Oct 14. Chemistry. 2015. PMID: 26462463
-
Model studies on a carprofen derivative as dual photosensitizer for thymine dimerization and (6-4) photoproduct repair.Chembiochem. 2007 Mar 5;8(4):402-7. doi: 10.1002/cbic.200600394. Chembiochem. 2007. PMID: 17285658
-
Photosensitized splitting of thymine dimer or oxetane unit by a covalently N-linked carbazole via electron transfer in different marcus regions.J Phys Chem B. 2010 Aug 5;114(30):9827-32. doi: 10.1021/jp1035579. J Phys Chem B. 2010. PMID: 20614917
-
Photosensitized Dimerization of Tyrosine: The Oxygen Paradox.Photochem Photobiol. 2022 May;98(3):687-695. doi: 10.1111/php.13557. Epub 2021 Nov 17. Photochem Photobiol. 2022. PMID: 34738644 Review.
-
Photosensitized generation of singlet oxygen.Photochem Photobiol. 2006 Sep-Oct;82(5):1161-77. doi: 10.1562/2006-03-03-IR-833. Photochem Photobiol. 2006. PMID: 16683906 Review.
Cited by
-
Reductive Photocycloreversion of Cyclobutane Dimers Triggered by Guanines.J Org Chem. 2023 Jul 21;88(14):10111-10121. doi: 10.1021/acs.joc.3c00930. Epub 2023 Jul 12. J Org Chem. 2023. PMID: 37437138 Free PMC article.
References
-
- Cadet J.; Voituriez L.; Hruska F. E.; Kan L. S.; de Leeuw F. A. A. M.; Altona C. Characterization of Thymidine Ultraviolet Photoproducts. Cyclobutane Dimers and 5,6-Dihydrothymidines. Can. J. Chem. 1985, 63, 2861–2868. 10.1139/v85-477. - DOI
-
- Sauvaigo S.; Douki T.; Odin F.; Caillat S.; Ravanat J.-L.; Cadet J. Analysis of Fluoroquinolone-Mediated Photosensitization of 2′-Deoxyguanosine, Calf Thymus and Cellular DNA: Determination of Type-I, Type-II and Triplet–Triplet Energy Transfer Mechanism Contribution¶. Photochem. Photobiol. 2001, 73, 230–237. 10.1562/0031-8655(2001)0730230AOFMPO2.0.CO2. - DOI - PubMed
-
- Noonan F. P.; Zaidi M. R.; Wolnicka-Glubisz A.; Anver M. R.; Bahn J.; Wielgus A.; Cadet J.; Douki T.; Mouret S.; Tucker M. A.; Popratiloff A.; Merlino G.; de Fabo E. C. Melanoma Induction by Ultraviolet A but Not Ultraviolet B Radiation Requires Melanin Pigment. Nat. Commun. 2012, 3, 884.10.1038/ncomms1893. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

