In vivo degradation forms, anti-degradation strategies, and clinical applications of therapeutic peptides in non-infectious chronic diseases
- PMID: 35981605
- PMCID: PMC9687107
- DOI: 10.1016/j.ejphar.2022.175192
In vivo degradation forms, anti-degradation strategies, and clinical applications of therapeutic peptides in non-infectious chronic diseases
Abstract
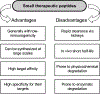
Current medicinal treatments for diseases comprise largely of two categories: small molecular (chemical) (e.g., aspirin) and larger molecular (peptides/proteins, e.g., insulin) drugs. Whilst both types of therapeutics can effectively treat different diseases, ranging from well-understood (in view of pathogenesis and treatment) examples (e.g., flu), to less-understood chronic diseases (e.g., diabetes), classical small molecule drugs often possess significant side-effects (a major cause of drug withdrawal from market) due to their low- or non-specific targeting. By contrast, therapeutic peptides, which comprise short sequences from naturally occurring peptides/proteins, commonly demonstrate high target specificity, well-characterized modes-of-action, and low or non-toxicity in vivo. Unfortunately, due to their small size, linear permutation, and lack of tertiary structure, peptidic drugs are easily subject to rapid degradation or loss in vivo through chemical and physical routines, thus resulting in a short half-life and reduced therapeutic efficacy, a major drawback that can reduce therapeutic efficiency. However, recent studies demonstrate that the short half-life of peptidic drugs can be significantly extended by various means, including use of enantiomeric or non-natural amino acids (AAs) (e.g., L-AAs replacement with D-AAs), chemical conjugation [e.g., with polyethylene glycol], and encapsulation (e.g., in exosomes). In this context, we provide an overview of the major in vivo degradation forms of small therapeutic peptides in the plasma and anti-degradation strategies. We also update on the progress of small peptide therapeutics that are either currently in clinical trials or are being successfully used in clinical therapies for patients with non-infectious diseases, such as diabetes, multiple sclerosis, and cancer.
Keywords: Anti-degradation strategies; Clinical applications; In vivo degradation forms; Therapeutic peptide.
Copyright © 2022 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest.
RGG holds stock in and is a company officer at the Tiny Cargo Company Inc., which has licensed exosomal technology from Virginia Tech. RGG is also a non-remunerated scientific advisory board member and stockholder of FirstString Research Inc, which licensed αCT1 peptide from the Medical University of South Carolina.
Figures

Similar articles
-
Biomedical Applications of a Novel Class of High-Affinity Peptides.Acc Chem Res. 2021 Sep 21;54(18):3576-3592. doi: 10.1021/acs.accounts.1c00239. Epub 2021 Aug 18. Acc Chem Res. 2021. PMID: 34406761
-
Enantiomeric purity of synthetic therapeutic peptides: A review.Chirality. 2024 Mar;36(3). doi: 10.1002/chir.23652. Chirality. 2024. PMID: 38448043 Review.
-
Peptide chemistry toolbox - Transforming natural peptides into peptide therapeutics.Bioorg Med Chem. 2018 Jun 1;26(10):2759-2765. doi: 10.1016/j.bmc.2018.01.012. Epub 2018 Jan 31. Bioorg Med Chem. 2018. PMID: 29395804 Review.
-
Design Strategies and Medicinal Applications of Metal-Peptidic Bioconjugates.Bioconjug Chem. 2020 May 20;31(5):1279-1288. doi: 10.1021/acs.bioconjchem.0c00152. Epub 2020 Apr 14. Bioconjug Chem. 2020. PMID: 32243752 Review.
-
Peptides and peptide conjugates: therapeutics on the upward path.Future Med Chem. 2012 Aug;4(12):1567-86. doi: 10.4155/fmc.12.76. Future Med Chem. 2012. PMID: 22917246 Review.
Cited by
-
GnRH Peptide Antagonist: Comparative Analysis of Chemistry and Formulation with Implications for Clinical Safety and Efficacy.Pharmaceuticals (Basel). 2024 Dec 31;18(1):36. doi: 10.3390/ph18010036. Pharmaceuticals (Basel). 2024. PMID: 39861098 Free PMC article. Review.
-
Quantifying and Controlling the Proteolytic Degradation of Cell Adhesion Peptides.ACS Biomater Sci Eng. 2024 Aug 12;10(8):4916-4926. doi: 10.1021/acsbiomaterials.4c00736. Epub 2024 Jul 5. ACS Biomater Sci Eng. 2024. PMID: 38968389 Free PMC article.
-
A Streamlined High-Throughput LC-MS Assay for Quantifying Peptide Degradation in Cell Culture.J Biomed Mater Res A. 2025 Jan;113(1):e37864. doi: 10.1002/jbm.a.37864. J Biomed Mater Res A. 2025. PMID: 39806927
-
A Streamlined High-Throughput LC-MS Assay for Quantifying Peptide Degradation in Cell Culture.bioRxiv [Preprint]. 2024 Oct 15:2024.10.11.617883. doi: 10.1101/2024.10.11.617883. bioRxiv. 2024. Update in: J Biomed Mater Res A. 2025 Jan;113(1):e37864. doi: 10.1002/jbm.a.37864. PMID: 39463983 Free PMC article. Updated. Preprint.
-
Quantifying and controlling the proteolytic degradation of cell adhesion peptides.bioRxiv [Preprint]. 2024 Apr 24:2024.04.19.590329. doi: 10.1101/2024.04.19.590329. bioRxiv. 2024. Update in: ACS Biomater Sci Eng. 2024 Aug 12;10(8):4916-4926. doi: 10.1021/acsbiomaterials.4c00736. PMID: 38712239 Free PMC article. Updated. Preprint.
References
-
- Adjei A, Doyle R, Pratt M, Finley R, Johnson E, 1990. Bioavailability of leuprolide following intratracheal administration to beagle dogs. Int. J. Pharm 61, 135–144. 10.1016/0378-5173(90)90052-6. - DOI
-
- Agerso H, Seiding Larsen L, Riis A, Lovgren U, Karlsson MO, Senderovitz T, 2004. Pharmacokinetics and renal excretion of desmopressin after intravenous administration to healthy subjects and renally impaired patients. Br. J. Clin. Pharmacol 58, 352–358. 10.1111/j.1365-2125.2004.02175.x. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

