BTZ-Derived Benzisothiazolinones with In Vitro Activity against Mycobacterium tuberculosis
- PMID: 35982823
- PMCID: PMC9380706
- DOI: 10.1021/acsmedchemlett.2c00215
BTZ-Derived Benzisothiazolinones with In Vitro Activity against Mycobacterium tuberculosis
Abstract
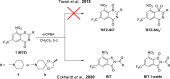
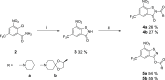
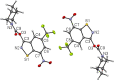
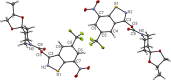
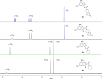
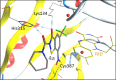
8-Nitro-1,3-benzothiazin-4-ones (BTZs) are known as potent antitubercular agents. BTZ043 as one of the most advanced compounds has reached clinical trials. The putative oxidation products of BTZ043, namely, the corresponding BTZ sulfoxide and sulfone, were reported in this journal (Tiwari et al. ACS Med. Chem Lett. 2015, 6, 128-133). The molecular structures were later revised to the constitutionally isomeric benzisothiazolone and its 1-oxide, respectively. Here, we report two BTZ043-derived benzisothiazolinones (BITs) with in vitro activity against mycobacteria. The constitutionally isomeric O-acyl benzisothiazol-3-ols, in contrast, show little or no antimycobacterial activity in vitro. The structures of the four compounds were investigated by X-ray crystallography and NMR spectroscopy. Molecular covalent docking of the new compounds to Mycobacerium tuberculosis decaprenylphosphoryl-β-d-ribose 2'-epimerase (DprE1) suggests that the active BITs exert antimycobacterial activity through inhibition of DprE1 like BTZs.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




Similar articles
-
Crystallographic evidence for unintended benzisothiazolinone 1-oxide formation from benzothiazinones through oxidation.Acta Crystallogr C Struct Chem. 2020 Sep 1;76(Pt 9):907-913. doi: 10.1107/S2053229620010931. Epub 2020 Aug 21. Acta Crystallogr C Struct Chem. 2020. PMID: 32887862 Free PMC article.
-
Synthesis, structural characterization and antimycobacterial evaluation of several halogenated non-nitro benzothiazinones.Med Chem Res. 2021;30(8):1523-1533. doi: 10.1007/s00044-021-02735-4. Epub 2021 Jun 10. Med Chem Res. 2021. PMID: 34131377 Free PMC article.
-
Syntheses and Antituberculosis Activity of 1,3-Benzothiazinone Sulfoxide and Sulfone Derived from BTZ043.ACS Med Chem Lett. 2014 Nov 29;6(2):128-33. doi: 10.1021/ml5003458. eCollection 2015 Feb 12. ACS Med Chem Lett. 2014. PMID: 25699139 Free PMC article.
-
Decaprenylphosphoryl-β-D-ribose 2'-epimerase from Mycobacterium tuberculosis is a magic drug target.Curr Med Chem. 2010;17(27):3099-108. doi: 10.2174/092986710791959693. Curr Med Chem. 2010. PMID: 20629622 Review.
-
Current Affairs, Future Perspectives of Tuberculosis and Antitubercular Agents.Indian J Tuberc. 2018 Jan;65(1):15-22. doi: 10.1016/j.ijtb.2017.08.011. Epub 2017 Aug 14. Indian J Tuberc. 2018. PMID: 29332642 Review.
Cited by
-
Pharmacokinetics and Efficacy of the Benzothiazinone BTZ-043 against Tuberculous Mycobacteria inside Granulomas in the Guinea Pig Model.Antimicrob Agents Chemother. 2023 Apr 18;67(4):e0143822. doi: 10.1128/aac.01438-22. Epub 2023 Mar 28. Antimicrob Agents Chemother. 2023. PMID: 36975792 Free PMC article.
-
Crystal structure and anti-mycobacterial evaluation of 2-(cyclo-hexyl-meth-yl)-7-nitro-5-(tri-fluoro-meth-yl)benzo[d]iso-thia-zol-3(2H)-one.Acta Crystallogr E Crystallogr Commun. 2023 Nov 30;79(Pt 12):1194-1198. doi: 10.1107/S2056989023010137. eCollection 2023 Dec 1. Acta Crystallogr E Crystallogr Commun. 2023. PMID: 38313133 Free PMC article.
References
-
- Global Tuberculosis Report; World Health Organization: Geneva, Switzerland, 2021.
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

